Impact of Single Nucleotide Polymorphism in the ANKRD55 Gene on Occurrence and Clinical Characteristics of Rheumatoid Arthritis
-
Salehi, Rasoul
-
Pediatric Inherited Diseases Research Center, Research Institute for Primordial Prevention of Non-Communicable Disease and Department of Genetics and Molecular Biology, School of Medicine, Isfahan University of Medical Sciences, Isfahan, Iran
-
Motaghi, Mina
-
Division of Rheumatology, Department of Internal Medicine, School of Medicine, Isfahan University of Medical Sciences, Isfahan, Iran
-
Salehi, Amirhossein
-
Division of Rheumatology, Department of Internal Medicine, School of Medicine, Isfahan University of Medical Sciences, Isfahan, Iran
-
Karimzadeh, Hadi
-
Division of Rheumatology, Department of Internal Medicine, School of Medicine, Isfahan University of Medical Sciences, Isfahan, Iran
-
 Pakzad, Bahram
Division of Rheumatology, Department of Internal Medi-cine, School of Medicine, Isfahan University of Medical Science, Isfahan, Iran, Bpakzadd@yahoo.com
Pakzad, Bahram
Division of Rheumatology, Department of Internal Medi-cine, School of Medicine, Isfahan University of Medical Science, Isfahan, Iran, Bpakzadd@yahoo.com
-
Division of Rheumatology, Department of Internal Medicine, School of Medicine, Isfahan University of Medical Sciences, Isfahan, Iran
Abstract: Background: Rheumatoid Arthritis (RA) has multifactorial etiology and numerous genetic and environmental factors have been related to an increased risk of RA. Recently, Genome-Wide Association Studies (GWAS) suggested a large number of Single Nucleotide Polymorphisms (SNPs) loci affecting the susceptibility to RA. One of these loci is rs6859219 (C>A), a functional polymorphism in the ANKRD55 gene which was associated with the expression of ANKRD55 and IL6ST. In the current study, we evaluated the possible association between rs6859219 (intronic variant) in the ANKRD55 gene with RA risk in the Iranian population.
Methods: A case-control study using 118 RA patients and 115 healthy counterparts was undertaken in order to determine rs6859219 genotypes using real‑time polymerase chain reaction High‑Resolution Melting (HRM) method.
Results: There was a significant difference in the genotype and allele frequencies of rs6859219 between patients and controls (p<0.001). Logistic regression analysis demonstrates that CC genotype and C allele increased the risk of RA (OR for CC genotype= 7.12; 95%CI [3.51-15.05]/ OR for C allele=4.16; 95%CI [2.78-6.28]). Furthermore, regarding the dominant and recessive model of inheritance, RA patients indicated obvious association of the rs6859219 variant compared to healthy controls (p<0.001). Moreover, in the patient group, there was a significant correlation between C-Reactive Protein (CRP) concentration with rs6859219 polymorphism (p<0.001).
Conclusion: Our findings propose a substantial correlation between rs6859219 polymorphism and RA risk and clinical characteristics of this disease in the Iranian population.
Introduction :
Rheumatoid Arthritis (RA) has multifactorial etiology and numerous genetic (50-60%) and environmental factors have been related to an increased risk of RA 1. Several studies have shown that genetic factors are critically involved in the incidence of RA. For instance, twin studies have shown an increased rate of RA in monozygotic versus dizygotic twins and the heritability of this disease has been estimated to be at around 60% 2. Single Nucleotide Polymorphisms (SNPs) are the most common form of allelic variation in the human genome and have been recognized on average every 300 nucleotides on usual with a Minor Allele Frequency (MAF) greater than 1% 3,4.
With recent advances in genotyping, Genome-Wide Association Studies (GWAS) and SNP arrays have revealed several RA susceptibility loci. For example in one GWAS in European and Asian populations revealed more than 110 susceptibility loci for RA 5. Based on several previous GWAS studies, the ankyrin repeat domain-55 (ANKRD55) gene including intronic SNPs associated with the risk of autoimmune diseases such as RA, Multiple Sclerosis (MS), and juvenile idiopathic arthritis 6-8. One of the important polymorphisms in this gene is rs6859219 (C>A) which is located on 5q11.2. Some studies reported that rs6859219 is associated with expression levels of the ANKRD55 and, in details, risk alleles of this SNP are associated with higher levels of ANKRD55 in CD4+T cells; while, it does not seem to exert functional effects in terms of splicing and transcription factor binding site modification 8,9.
Nevertheless, until now the biological function of ANKRD55 is unknown and studies only referred to the interaction with numerous proteins by ankyrin repeat domains 10,11. IL6ST gene encodes the common cytokine receptor gp130. The receptor systems for IL6, LIF, OSM, CNTF, IL11, CTF1, and BSF3 can utilize IL6ST for initiating signal transmission. By way of example, binding of IL6 to IL6R induces IL6ST homodimerization and formation of a high-affinity receptor complex which activates Janus kinases, leading to the phosphorylation of IL6ST tyrosine residues and finally activation of STAT3 12-14. Therefore, dysregulation in IL6, IL6ST, and STAT3 pathways could result in an autoimmune condition 15. In the present study, for the first time, we evaluated the probable relationship between rs6859219 polymorphism with the risk of RA in the Iranian population.
Materials and Methods :
Study population
A total of 118 unrelated subjects with RA as a case group and 115 unrelated healthy subjects as a control group were included in this case-control study. Subjects in the case group were recruited from the Alzahra Hospital, Isfahan, Iran. All the RA patients met the diagnostic criteria created by the American College of Rheumatology (ACR) 16. Controls were also selected from the same population with no signs and personal and family history of RA or other immunological and autoimmune conditions. The study was approved by the university ethics board and all participants gave written informed consent. To evaluate any established risk factors for RA, all participants were asked to fill up a questionnaire in order to register the parameters known to influence the RA susceptibility risk including on sex, age (at sampling time) and age of onset, Body Mass Index (BMI, calculated as weight [kg] divided by height [m] squared), blood pressure, the presence of family history of RA and other autoimmune conditions were obtained using a structured questionnaire. Also, we recorded laboratory characteristics such as Erythrocyte Sedimentation Rate (ESR), C-Reactive Protein (CRP), White Blood Cell (WBC), hemoglobin, Platelet count test (PLT), creatinine, Blood Urea Nitrogen (BUN), Fasting Blood Sugar (FBS), High-Density Lipoprotein (HDL), Low-Density Lipoprotein (LDL), and Triglyceride (TG).
Genotyping of polymorphism
Approximately, 3 ml of the blood sample was collected into EDTA anticoagulant tubes from each contributor and stored at -20°C for DNA isolation. DNA was extracted using a DNA isolation kit (GeNet Bio; Korea) consistent with the instruction manual. The purity and concentration of extracted DNA were assessed by agarose gel electrophoresis and spectrophotometry, respectively, and then DNA was stored at
-20°C until genotyping by real‑time polymerase chain reaction High‑Resolution Melting (HRM) method.
The HRM method was used to determine rs6859219 polymorphism genotypes. The details of the HRM method were described in our previous studies 17,18. Amplification of fragment (141 bp) was performed using the primer sense (CGCTACAGTGGTGACCCC) and antisense (GTCATCTCCACCTGCCCATA). A 35-cycle PCR was carried out with the following conditions: 5 min at 95°C for denaturation of the template DNA for the first cycle, denaturation at 95°C for 20 s, annealing at 60°C for 30 s, and extension at 72°C for 20 s. In the HRM phase, the Rotor-Gene 6000™ measured the fluorescence in each 0.1°C/s temperature which was increased in the range of 65°C and 95°C. The melting curve was produced by the reduction in fluorescence with the increase in the temperature; and in analysis, nucleotide changes result in different curve patterns.
Statistical analyses
The SPSS 22 (IBM, Armonk, NY: IBM Corp) was used for statistical analyses. The allele and genotype frequencies of rs6859219 were tested for Hardy-Weinberg equilibrium by the χ2 test. Logistic regression analysis was accomplished to investigate the association between genotypes and RA and calculate specific Odds Ratios (ORs), 95% Confidential Intervals (CIs), and p-values. For demographic, clinical, and laboratory characteristics, p-values were calculated using independent sample t‑test, Chi‑square, or Mann–Whitney test. The significance level was set at p<0.05.
Study population
A total of 118 unrelated subjects with RA as a case group and 115 unrelated healthy subjects as a control group were included in this case-control study. Subjects in the case group were recruited from the Alzahra Hospital, Isfahan, Iran. All the RA patients met the diagnostic criteria created by the American College of Rheumatology (ACR) 16. Controls were also selected from the same population with no signs and personal and family history of RA or other immunological and autoimmune conditions. The study was approved by the university ethics board and all participants gave written informed consent. To evaluate any established risk factors for RA, all participants were asked to fill up a questionnaire in order to register the parameters known to influence the RA susceptibility risk including on sex, age (at sampling time) and age of onset, Body Mass Index (BMI, calculated as weight [kg] divided by height [m] squared), blood pressure, the presence of family history of RA and other autoimmune conditions were obtained using a structured questionnaire. Also, we recorded laboratory characteristics such as Erythrocyte Sedimentation Rate (ESR), C-Reactive Protein (CRP), White Blood Cell (WBC), hemoglobin, Platelet count test (PLT), creatinine, Blood Urea Nitrogen (BUN), Fasting Blood Sugar (FBS), High-Density Lipoprotein (HDL), Low-Density Lipoprotein (LDL), and Triglyceride (TG).
Genotyping of polymorphism
Approximately, 3 ml of the blood sample was collected into EDTA anticoagulant tubes from each contributor and stored at -20°C for DNA isolation. DNA was extracted using a DNA isolation kit (GeNet Bio; Korea) consistent with the instruction manual. The purity and concentration of extracted DNA were assessed by agarose gel electrophoresis and spectrophotometry, respectively, and then DNA was stored at
-20°C until genotyping by real‑time polymerase chain reaction High‑Resolution Melting (HRM) method.
The HRM method was used to determine rs6859219 polymorphism genotypes. The details of the HRM method were described in our previous studies 17,18. Amplification of fragment (141 bp) was performed using the primer sense (CGCTACAGTGGTGACCCC) and antisense (GTCATCTCCACCTGCCCATA). A 35-cycle PCR was carried out with the following conditions: 5 min at 95°C for denaturation of the template DNA for the first cycle, denaturation at 95°C for 20 s, annealing at 60°C for 30 s, and extension at 72°C for 20 s. In the HRM phase, the Rotor-Gene 6000™ measured the fluorescence in each 0.1°C/s temperature which was increased in the range of 65°C and 95°C. The melting curve was produced by the reduction in fluorescence with the increase in the temperature; and in analysis, nucleotide changes result in different curve patterns.
Statistical analyses
The SPSS 22 (IBM, Armonk, NY: IBM Corp) was used for statistical analyses. The allele and genotype frequencies of rs6859219 were tested for Hardy-Weinberg equilibrium by the χ2 test. Logistic regression analysis was accomplished to investigate the association between genotypes and RA and calculate specific Odds Ratios (ORs), 95% Confidential Intervals (CIs), and p-values. For demographic, clinical, and laboratory characteristics, p-values were calculated using independent sample t‑test, Chi‑square, or Mann–Whitney test. The significance level was set at p<0.05.
Results :
In our study, we investigated a total of 118 patients (35 males and 83 females with a mean age of 47.3983±9.801 years) and 115 control (37 males and 78 females with a mean age of 46.0304±12.430 years) for ANKRD55 (rs6859219) polymorphism. The distributions of selected characteristics of the cases and controls are presented in table 1. There was no substantial correlation between case and control groups regarding age (p=0.238) and sex (p=0.678), demonstrating that for these variables matching was adequate. Between the two groups of subjects, there was a significant difference in terms of BMI (p<0.001) and just 19 patients had positive family history of RA and other autoimmune conditions. Based on laboratory tests, the WBC count and concentration of ESR, CRP, and creatinine was significantly higher in patients than in healthy controls (p<0.05). On the other hand, the level of hemoglobin was significantly lower in patients than in healthy controls (p<0.001). The detailed laboratory characteristics of patients with RA and healthy controls are listed in table 2.
Genotype and allele distribution
The genotype distribution of rs6859219 polymorphism in cases and control groups was in agreement with Hardy-Weinberg equilibrium. There was a significant difference in the genotype frequencies of rs6859219 between RA patients and controls. The frequencies of AA, AC, CC genotypes in the control group were 57, 28, and 15.0%, respectively, and the genotype frequencies in the case group were 30.0, 15.0, and 55.0%, respectively. Significant association was found between CC [compared with AA; (p<0.001)] genotype and RA risk. Our assessments on various models of inheritance for rs6859219 polymorphism established that the genotype frequency of this polymorphism was meaningfully different under the dominant and recessive model among RA subjects and controls group (p<0.001). In allele distribution analysis, we found an increased level of allele C in patients (63%) compared with controls (29%) and this allele was associated with an increased risk of RA (p<0.001) (Table 3). Our analysis established that stratification based on mean serum concentration of CRP in the patient group is significantly different in genotype stratification (p<0.001). In detail, patients with risk allele (C) have a higher concentration of CRP (Table 4).
Discussion :
To the best of our knowledge, our study is the first research in the Iranian population that evaluated the association between ANKRD55 polymorphism, rs-6859219, with the RA risk. Many GWAS suggested numerous different SNPs loci in different genes associated with RA. The rs6859219 polymorphism in the ANKRD55 gene is one of these loci which GWAS revealed its association with RA 7. This polymorphism (rs6859219) is located in the seventh intron of the ANKRD55 gene 8. This gene highly expressed in CD4+T cells but the biological function of ANKRD55 is unknown. Lapuente et al demonstrated that rs-6859219 is associated with higher levels of ANKRD55 in CD4+T cells. Also, Clark and coworkers demonstrated that the location of this variant acts as an enhancer for IL6ST and the risk allele in this variant was associated with modulation of IL6ST expression 19. Some studies referred to the influence of this polymorphism in autoimmune disease susceptibility through an effect on the expression of IL6ST 7. In the current study, logistic regression analysis determined that homozygous CC genotypes compared with the AA genotype increases the risk of RA [(CC vs. AA; OR=7.12 (3.51-15.05)]. Similarly, the difference model of inheritance increases the risk of disease (OR for recessive and dominant inheritance were 7.00 and for 3.17, respectively). Furthermore, subjects with allele C were more frequently affected with RA than subjects with A allele [OR=4.16; 95%CI (2.78-6.28)] (Table 3). Our finding was consistent with a GWAS study in the European descent that carried out by Stahl et al in 2010 which demonstrated for the first time the association of this polymorphism with RA 7. In another study, Lill and coworkers in their GWAS studies carried out on samples from Germany and France revealed that this polymorphism also is correlated with increased risk of MS. Likewise, the other study on the Spanish population demonstrated that rs6859219 is associated with MS. Yazdanpanah et al reported that CC genotype in this polymorphism was correlated with increased risk of MS in Iranian population 20.
Furthermore, in the patient group, we found a significant correlation between CRP concentration and rs6859219 polymorphism (p<0.001) (Table 4). The amount of these factors indicates levels of inflammation in the body and refers to active disease. This result demonstrates the association of risk allele with the severity of the disease 21,22.
Conclusion :
Our analysis showed that rs6859219 is a strong determinant for RA risk and disease activity. However, performing replicative studies in every population is a necessity to validate these results. Finally, in this work, probably, some possible limitations in the statistical validity of our results such as small population size exist; so further association studies in larger sample size would help to confirm the suggested correlations. Also, other polymorphisms that were not included in our study might be involved in determining the risk of RA, thus future studies are necessary.
Acknowledgement :
We would like to appreciate the financial support provided by Isfahan University of Medical Sciences.
Conflict of Interest :
None declared.
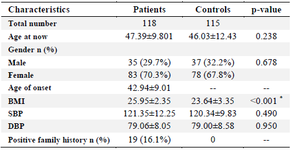
Table 1. Baseline characteristics of RA patients and control subjects participated in the study
RA: Rheumatoid Arthritis, BMI: Body Mass Index, SBP: Systolic Blood Pressure, DBP: Diastolic Blood Pressure, SD: Standard deviation.
Data are mean±SD, or n (%). * p<0.05.
|
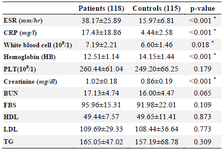
Table 2. Laboratory characteristics of patients with RA and controls group
RA: Rheumatoid Arthritis, ESR: Erythrocyte Sedimentation Rate, CRP: C‑ reactive Protein, BUN: Blood Urea Nitrogen, PLT: Platelet, HDL: High‑ Density Lipoprotein, LDL: Low‑Density Lipoprotein, TG: Triglyceride, FBS: Fasting Blood Sugar, SD: Standard Deviation.
Data are mean±SD, or n (%). * p<0.05.
|
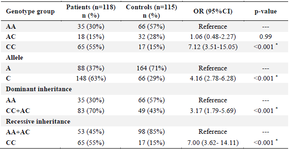
Table 3. Association between genotypes and allele frequency with RA risk
* p<0.05; RA: Rheumatoid arthritis.
|
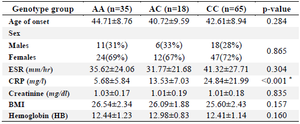
Table 4. Stratification analyzes of the ANKRD55 polymorphism (rs6859219) in patients (118 RA patients)
ESR: Erythrocyte Sedimentation Rate, CRP:C‑reactive Protein, BMI: Body Mass Index, SD: Standard Deviation.
Data are mean±SD, or n (%). * p<0.05.
|
|