Targeted Overexpression of NDRG2 using Survivin Promoter Reduces Viability and Invasiveness of A549 Cell Line
-
Fanian, Maryam
-
Division of Medical Biotechnology, Department of Laboratory Sciences, School of Paramedical Sciences, Shiraz University of Medical Sciences, Shiraz, Iran
-
Department of Infectious Diseases and Public Health, City University of Hong Kong, Kowloon, Hong Kong SAR, China
-
Rafiei , Gholamreza
-
Diagnostic Laboratory Sciences and Technology Research Center, School of Paramedical Sciences, Shiraz University of Medical Sciences, Shiraz, Iran
-
Alizadeh Zarei, Marzieh
-
Division of Medical Biotechnology, Department of Laboratory Sciences, School of Paramedical Sciences, Shiraz University of Medical Sciences, Shiraz, Iran
-
 Takhshid, Mohammad Ali
Division of Medical Biotechnology, Department of Laboratory Sciences, School of Paramedical Sciences, Shiraz University of Medical Sciences, Shiraz, Iran , Tel: +98 0917 3121699; E-mail: takhshidma@sums.ac.ir
Takhshid, Mohammad Ali
Division of Medical Biotechnology, Department of Laboratory Sciences, School of Paramedical Sciences, Shiraz University of Medical Sciences, Shiraz, Iran , Tel: +98 0917 3121699; E-mail: takhshidma@sums.ac.ir
-
Diagnostic Laboratory Sciences and Technology Research Center, School of Paramedical Sciences, Shiraz University of Medical Sciences, Shiraz, Iran, Shiraz, Iran
Abstract: Background: Anti-tumor effects of N-myc Downstream Regulated Gene2 (NDRG2) have been demonstrated in many tumors. In the present study, NDRG2 was specifically overexpressed in lung cancer cell line using Survivin Promoter (Sur-P). Then, the effects of NDRG2 overexpression on viability, apoptosis, migration, and invasion of A549 cells were evaluated.
Methods: Recombinant pAdenoVator-Sur-P-NDRG2-IRES-GFP plasmid harboring NDRG2 gene under transcriptional control of Sur-P and mock plasmid were constructed. A549 lung tumor cells and LX-2 cells (non-tumor cell line) were transfected with pAdenoVator-Sur-P-NDRG2-IRES-GFP, pAdenoVator-CMV-NDRG2-IRES-GFP, or mock plasmids. Tumor specificity of Sur-P was evaluated using fluorescent microscopy for GFP expression. The effects of NDRG2 overexpression on cell viability, apoptosis, and migration of A549 cells were measured using MTT, annexinV/7-AAD flow cytometry, and transwell migration assay, respectively. NDRG2 and matrix metalloproteinase-2 (MMP-2) expression were measured using real time- PCR.
Results: pAdenoVator-Sur-P-NDRG2-IRES-GFP transfection resulted in a huge GFP expression in A549 cells, but not in LX-2 cells. The results of real time-PCR analysis also showed that pAdenoVator-Sur-P-NDRG2-IRES-GFP transfection led to an abundant NDRG2 expression in A549 cells. NDRG2 overexpression decreased A549 cell viability through increasing cell apoptosis. In addition, migration, invasion, and MMP-2 expression decreased following NDRG2 overexpression in A549 cells.
Conclusion: The findings indicate that the targeted overexpression of NDRG2 using Sur-P can reduce the viability and invasiveness of A549 cells, suggesting possible benefits of this approach in lung cancer therapy.
Introduction :
According to GLOBOCAN 2020 statistics, lung cancer is the second in incidence (11.4%) and the first in mortality rate (18%) among 36 prevalent cancers worldwide 1. Histologically, lung cancer is classified as Small-Cell Lung Carcinoma (SCLC) and Non-SCLC (NSCLC), which account for about 15% and 85% of lung cancer cases, respectively 2. Surgical resection, radiotherapy, chemotherapy, targeted therapy, and combination therapy are available therapeutic strategies for lung cancer. Four classes of chemotherapeutic drugs are used for the treatment of patients with NSCLC which include alkylating agents (cisplatin and carboplatin) microtubule-targeting drugs (paclitaxel and docetaxel), antimetabolites (pemetrexed and gemcitabine), and topoisomerase inhibitors (etoposide) 3. Although chemotherapy still plays an important role in the treatment of patients with lung cancer, drug toxicity and resistance are the main challenges in this issue. Previous studies have revealed the role several mechanisms in developing drug resistance including reduced drug uptake to tumor cells, increased drug efflux from the tumor cells, genetics and epigenetic modifications, apoptosis dysfunction, and acquisition of cancer stem cell-like properties 4. Therefore, finding an efficient and safe therapeutic method that specifically targets cancer cells has been the focus of many studies. In this context, gene therapy has emerged as a promising approach 5.
Tumor Specific Promoters (TSPs), transcriptional regulatory elements that ensure targeted and efficient expression of genes in tumor cells, are key elements in the success of gene therapeutic approaches 6. Survivin, an apoptosis inhibitory protein, is expressed in numerous types of cancer including SCLC and NSCLC 7. Numerous studies have revealed tumor specificity and strength of Survivin promoter (Sur-P) in the expression of various genes in several tumor cells 8. Moreover, its transcriptional activity is increased in hypoxia, a condition that usually found in tumor cells 9. Specific overexpression of SP1 protein, a transcription factor that directly binds to Sur-P, results in high transcriptional activity of Sur-P in lung tumor cells 10.
N-myc Downstream Regulated Gene 2 (NDRG2) is a multifunctional protein with crucial roles in the regulation of cell growth and differentiation. NDRG2 expression is downregulated in many cancers including lung cancer. Furthermore, its expression has been found to be inversely associated with tumor stage, tumor size, and invasion of tumor cells to circulation and directly associated with patient’s survival time 11,12. Moreover, Faraji et al showed that NDRG2 upregulation in A549 cells suppressed cell viability and invasion of tumor cells through decreasing expressions of Matrix Metalloproteinases-2 and -9 (MMP-2 and MMP-9) 13.
The present study aimed to construct a recombinant plasmid harboring NDRG2 cDNA under the transcriptional control of Sur-P to evaluate the tumor specificity and strength of this promoter in enhancing the NDRG2 expression. The results revealed specific overexpression of NDRG2 in A549 cell lines using this plasmid. Furthermore, NDRG2 overexpression was associated with increased apoptosis and decreased invasiveness of the tumor cells.
Materials and Methods :
Construction of plasmids: Three types of recombinant plasmids were used in this study including pAdenoVator-CMV-NDRG2-IRES-GFP (NDRG2 under transcriptional control of CMV promoter), pAdenoVator-Sur-P-NDRG2-IRES-GFP (NDRG2 under transcriptional control of Sur-P), and pAdenoVator-Sur-P-IRES-GFP (Mock plasmid). CMV-NDRG2 was constructed in our previous studies 24,26. To construct Sur-P-NDRG2 plasmid, Sur-P 14 was amplified from genomic DNA (prepared from peripheral blood mononuclear cells) using forward (5'-CCGGAGTCGACGTTCTTTGA AAGCAG-3'; contained SalI restriction site) and reverse (5'-TTAGCTCTAGACAACGTCGG GGCACC-3'; contained XbaI restriction site) primers. Sur-P fragment was digested by XbaI and SalI restriction enzymes (Thermo Scientific, USA) and cloned into the pAdenoVator- IRES-GFP vector using T4 DNA ligase (Thermo Scientific, USA). The ligation mixture was then transformed into competent Escherichia coli (E. coli) DH5α. The formation of Sur-P-NDRG2 was validated using colony PCR and sequencing.
Cells and cell culture conditions: A549 cell line (ATCC ref. No. CLL-185) was purchased from Pasture Institute, Iran. LX-2 (human hepatic stellate cell line) was kindly provided by Dr. Younes Hoseini. A549 cells and LX-2 cells were cultured in RPMI 1640 (Gibco/Invitrogen, Carlsbad, CA) supplemented with 10% and 5% Fetal Bovine Serum (FBS) (Gibco, USA) and 1% penicillin-streptomycin (Gibco, USA). Both cell lines were incubated at 37°C with 5% CO2 atmosphere.
Overexpression of the NDRG2 gene in A549 cells: To evaluate tumor specific activity of Sur-P, A549 and LX-2 cell lines were transfected with pAdenoVator-CMV-NDRG2-IRES-GFP, pAdenoVator-Sur-P-NDRG2-IRES-GFP, and mock plasmids using lipofectamine™ 2000 (Invitrogen, Carlsbad, CA) according to the manufacturer’s instructions. After 48 hr, the transfected cells were detached using 0.25% Trypsin-EDTA, washed, and resuspended in cold PBS buffer. Fluorescence microscopy and flow cytometry analyses were used to monitor the cellular expression of GFP and to measure the efficiency of transfection. Fluorescence microscopy was performed on an inverted microscope (Hund, Germany) with filter sets designed for GFP. Flow cytometric analysis was conducted using BD FACSCalibur flow cytometer (BD Biosciences, USA). For each sample, 10,000 events were collected. The expression of NDRG2 was evaluated using real-time PCR as described in the following section.
Cell viability assay using the MTT method: The effect of NDRG2 overexpression on cell viability was evaluated using MTT assay. Briefly, 6×103 A549 cells were seeded on each well of a 96-well plate and were transfected with either pAdenoVator-Sur-P-NDRG2-IRES-GFP plasmid (NDRG2 group) or mock plasmid (pAdenoVator-Sur-P-IRES-GFP) using Lipofectamine 2000 (Invitrogen, USA). Forty-eight hours after transfection, the cells were treated with 10 µl of MTT [3-(4,5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide] solution followed by a four-hour incubation. Afterwards, the media were removed, formazan crystals were dissolved by Dimethyl Sulfoxide (DMSO), and the absorbance was measured at 545 nm.
Cell apoptosis assay: Annexin V/7AAD flow cytometry was used to evaluate the effect of NDRG2 overexpression on cell apoptosis. In doing so, A549 cells were transfected with NDRG2 or mock plasmids, as described previously. Forty-eight hr after transfection, the cells were harvested using trypsin digestion (0.25%) and centrifugation. The cells were then washed with ice-cold PBS, suspended in 1× annexin V binding buffer, and stained with PE annexin V and 7-AAD at room temperature in the dark for 15 min. The percentages of live and apoptotic cells were determined using the FACSCalibur instrument (BD Bioscince.USA) and flowjo software analysis.
Migration and invasion assay: Transwell migration assay was applied using 24-well transwell insert and 8 µm pore filters (BD Bioscience, Bedford, MA) to evaluate the effects of NDRG2 overexpression on the migration of the transfected A549 cells. Briefly, 48 hr after transfection, A549 cells (5×104) were suspended with a serum-free medium, kept serum starved for 24 hr, transferred to the upper part of the transwell, and incubated at 37°C. The cells were then allowed to migrate to the lower part of the transwell, which was filled with RPMI supplemented with 10% FBS. After 24 hr incubation, the cells that migrated to the lower surface of the transwell were fixed with methanol for five minutes and were stained with 0.1% crystal violet. For invasion assays, the transwell was coated with 100 µl (1 mg/ml) matrigel (BD Bioscience, Bedford, MA). Forty-eight hr after transfection, A549 cells (5×104) were plated onto the upper part of the matrigel-coated transwell chamber and were incubated for 48 hr. The invaded cells were then fixed with methanol, stained, and counted. The number of invading or migrating cells was determined by counting five fields (400×) of a light microscope on each membrane and was calculated as the mean number of cells per field 15,16.
Real time polymerase chain reaction: A549 and LX-2 cell lines were cultured at 1×105 and 1.5×105 counts in six-well plates, respectively. Both cell lines were transfected with pAdenoVator-Sur-P-NDRG2-IRES-GFP. Forty-eight hr after transfection, the cells were exposed to 1 ml TRIzol (Thermo Fisher Scientific. USA) per 1×106 cells and the RNAs were extracted according to the manufacturer’s protocol. After that, cDNAs were synthesized from the extracted RNAs using Takara kit according to the manufacturer’s instructions. Quantitative real-time PCR was performed using specific primers (Table 1), SYBR-Green master mix, and the Rotor-Gene Q (Qiagen. Germany) in a total reaction volume of 20 μl.
Statistical analysis: All statistical analyses were performed using the SPSS software, version 15.0. Statistical differences between the study groups were analyzed using Mann-Whitney or Kruskal-Wallis test. The data represented mean ± Standard Deviation (SD) of at least three independent experiments. The p<0.05 was considered statistically significant.
Results :
Tumor specificity of survivin promoter: Tumor specific activity of Sur-P was evaluated using florescence microscopy (Figure 1 A-D). As illustrated in figure 1A, transfection of pAdenoVator-CMV-NDRG2-IRES-GFP plasmid into normal LX-2 cells led to a high GFP expression (Figure 1A), while transfection with pAdenoVator-Sur-P-NDRG2-IRES-GFP did not lead to GFP expression (Figure 1B). In A549 cells, however, transfection with both pAdenoVator-CMV-NDRG2-IRES-GFP (Figure 1C) and pAdenoVator-Sur-P-NDRG2-IRES-GFP (Figure 1D) led to GFP expression, suggesting tumor specificity of Sur-P. The results of real-time PCR also revealed an abundant NDRG2 expression in the A549 cells (1200-fold) transfected with the pAdenoVator-Sur-P-NDRG2-IRES-GFP plasmid compared to the control non-transfected cells, suggesting the high strength of Sur-P in inducing NDRG2 expression in A549 tumor cells.
Cell viability assay: Figure 2 shows the results of MTT assay. The data revealed that NDRG2 overexpression decreases the viability of A549 cells compared to the control non-transfected cells (p<0.001) and the mock plasmid-transfected group (p=0.002). No significant difference was observed between the viability of control and mock groups (p=0.243).
Flow cytometric analysis of apoptosis: As shown in figure 3, NDRG2 overexpression increased the percentage of total apoptotic cells compared to the mock-transfected (p=0.047) and control non-transfected cells (p=0.039). However, a decrease was detected in cell viability following NDRG2 overexpression compared to the mock and control non-transfected cells.
Migration and invasion assay: Figure 4 illustrates the effects of NDRG2 overexpression on the migration and invasion of the A549 cells. Based on the data presented in figure 4, NDRG2 overexpression reduced the number of migrated and invaded cells compared to the mock and control cells, suggesting the anti-metastatic effects of NDRG2 on A549 cells.
The effect of NDRG2 overexpression on the level of MMP-2 mRNA: The effect of NDRG2 overexpression on MMP-2 expression was evaluated using real-time PCR. The results revealed a significant decrease in the MMP-2 mRNA level (p=0.045) compared to the control non-transfected cells (Figure 5).
Discussion :
Several studies have revealed the beneficial effects of various gene therapy methods including inhibition of oncogenes and replacement of tumor-suppressor genes in the treatment of lung cancer 5. However, lack of tumor specificity is the most important limitation of these approaches. Two main strategies have suggested for increasing tumor specificity of gene therapy including targeted transfer to tumor cells via tumor specific receptors and targeted transcription of anti-tumor genes using TSPs. Despite the tumor specificity of various TSPs, low transcriptional efficiency usually hinders their utilization for gene therapy strategies. The present study assessed the specificity and efficiency of Sur-P in the overexpression of NDRG2 in A549 cells. The findings revealed an abundant expression of NDRG2 under the transcriptional control of Sur-P in the lung cancer cell line, but not in the normal cell line, compared to the CMV promoter that enhanced NDRG2 overexpression in both normal and tumor cells. Furthermore, NDRG2 overexpression decreased cell viability and invasiveness of A549 cells, suggesting specificity and efficiency of this strategy and its possible effectiveness in the treatment of NSCLC.
Survivin is anti-apoptotic protein that is specifically expressed in the most of human cancers including lung cancer. Numerous studies have revealed that efficient downregulation of survivin using transcriptional and post-translational approaches resulted in death and apoptosis of tumor cells and sensitized tumor cells to chemotherapeutic regimens 17. In addition, several studies have described the beneficial use of Sur-P for the targeted expression of anti-tumor genes 18. Bao et al used a 1092-bp fragment of the human Sur-P to control the expression of alkaline phosphatase in several tumor cell lines and reported its promoter activity in all evaluated tumor cells 19. However, the large size of the fragment hampered its use for the construction of recombinant plasmids 8. Therefore, Yang et al evaluated the efficiency of a number of surviving promoter fragments and indicated that a 269-bp fragment of Sur-P was sufficient for the specific and efficient expression of genes in human pancreatic and breast cancer cell lines 14. In the current study, we constructed a plasmid harboring Sur-P and human NDRG2 cDNA. Our data revealed that Sur-P can mediate NDRG2 overexpression in A549 cells, but not in LX-2 cells, suggesting the tumor specificity and strength of this promoter in lung cancer cells. LX-2 has been used in some studies to determine tumor specificity of some promoters 20,21. Nonetheless, LX-2 is a hepatic stellate cell line and the lack of evaluation of Sur-P activity in the lung epithelial cells is among the limitations of this study. Further studies are needed to determine transcriptional activity of Sur-P in the normal lung epithelial cells.
Previous studies have shown that suppressing expression of NDRG2 by genetic and epigenetic methods led to development of a variety of tumors including lung cancer. Li et al showed a decreased level of NDRG2 in human lung cancer tissues that was associated with the invasion and tumor size. In addition, 5-years survival time of patients with low NDRG2 expression was shorter than those with high NDRG2 expression 11. Thus, NDRG2 has become a target for cancer therapy, and numerous studies have revealed the potent effect of NDRG2 in reducing cell viability and invasiveness of tumor cells 22-26. Nevertheless, the role NDRG2 overexpression in lung cancer therapy has been investigated in a few studies 13. In the present study, human NDRG2 gene was specifically overexpressed using Sur-P in A549 cells. Consistent with the results of a previous study 13,27, our findings revealed that NDRG2 overexpression decreased viability and enhanced apoptosis of A549 cells. Several possible mechanisms can describe anti-tumor effects of NDRG2. In human colon carcinoma cells, it has been demonstrated that NDRG2 overexpression arrested cell cycle in G1 phase by downregulating cyclin D expression 28. Similarly, Kang et al evaluated the effects of NDRG2 overexpression on cell proliferation of ovarian cancer cells and showed that NDGR2 can suppress cell viability by increasing expression of apoptosis-related proteins. In addition, the authors revealed that NDRG2 overexpression significantly arrested cell cycle of ovarian cancer cell lines by reducing expression of cyclins 29. To our knowledge, the mechanisms of NDRG2 effects on the cell viability of lung tumor cells have not been investigated so far and further studies are needed to clarify the possible role cell cycle and apoptosis proteins in this issue.
The results of the current research also showed that NDRG2 overexpression remarkably reduced the invasion and migration of A549 cells, which was in agreement with the effects of NDRG2 on other tumor cells 23-25. MMPs are essential enzymes for the growth, invasion, and angiogenesis of lung tumor cells 30. MMP-2 (gelatinase A) is highly expressed in lung cancer and is correlated with the invasiveness of this malignancy 31. Findings of the current study revealed a decreased level of MMP-2 mRNA upon NDRG2 overexpression, indicating the possible role of this enzyme in the anti-invasive effects of NDRG2. This finding is line with the results of our previous study which showed the inhibitory effects of NDRG2 on MMP-2 enzymatic activity 13. Similarly, NDRG2 overexpression was shown to be associated with MMPs downregulation in renal cell carcinoma 32, prostate cancer 24, and melanoma cell lines 33. Several mechanisms may involve in suppressing effects of NDRG2 on MMPs expression, notably inhibition of nuclear factor kappa B activity 33 and phosphoinositide 3-kinase (PI3K)/AKT signaling pathway 34. Further investigations are required to determine the role these signaling pathways in the anti-metastatic effects of NDRG2 in lung cancer.
Conclusion :
The study findings suggested that Sur-P resulted in the specific overexpression of NDRG2 in lung tumor A549 cells. The results also exhibited increased cell death and apoptosis and decreased migration and invasion of A549 cells through inhibition of MMP-2 and MMP-9 expression. This strategy may be considered for the treatment of NSCLC. Yet, further in vivo studies are required to confirm the potential efficacy of this method in lung cancer therapy.
Acknowledgement :
This paper was a part of Maryam Fanian’s MSc thesis and was supported by grant No. 90-5971 from the Vice-Chancellor for Research Affairs of Shiraz University of Medical Sciences, Shiraz, Iran. The authors are grateful to all staff of the Diagnostic Laboratory Sciences and Technology Research Center and Shiraz Institute for Cancer Research Center for their technical assistance in this work. They would also like to appreciate Ms. A. Keivanshekouh at the Research Consultation Center (RCC) of Shiraz University of Medical Sciences for improving the use of English in the manuscript.
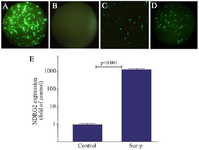
Figure 1. Tumor specificity of Green Fluorescent Protein (GFP) expression under Sur-P control. GFP was expressed in the LX-2 cells transfected with pAdenoVator-CMV-NDRG2-IRES-GFP (A), but not in those transfected with pAdenoVator-Sur-P-NDRG2-IRES-GFP(B). GFP was also expressed in the A549 cells transfected with both pAdenoVator-CMV-NDRG2-IRES-GFP (C) and pAdenoVator-Sur-P-NDRG2-IRES-GFP (D). Real-time PCR data (E) of NDRG2 expression in the A549 cells transfected with the pAdenoVator-Sur-P-NDRG2-IRES-GFP plasmid and control non-transfected cells. The data represent the mean±SD of at least three independent experiments.
|
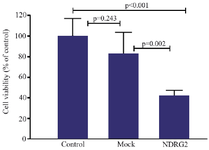
Figure 2. MTT assay results showing the effects of NDRG2 overexpression on A549 cell viability. A549 cells were transfected with pAdenoVator-Sur-p-NDRG2-IRES-GFP or mock plasmids and cell viability was evaluated using the MTT assay. The data were analyzed using Kruskal-Wallis test. The data represent the mean±SD of at least three independent experiments.
|
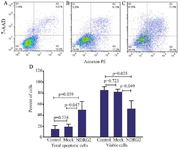
Figure 3. The effect of NDRG2 overexpression on the percentage of live and total apoptotic A549 cells. The charts show the percentages of live cells (Q4), early apoptotic cells (Q3), late apoptotic cells (Q2), and necrotic cells (Q1) in the control (A), mock (B), and NDRG2 (C) groups. The data (D) represent the mean±SD of three independent experiments. The data were analyzed using Kruskal-Wallis’s test.
|
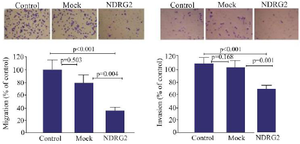
Figure 4. The effects of NDRG2 overexpression on the migration and invasion of A549 cells using transwell assay. The percentage of migrated and invaded A549 cells in the mock plasmid transfected (mock), pAdenoVator-Sur-P-NDRG2-IRES-GFP-transfected (NDR-G2), and control non-transfected cells. The data represent the mean± SD of three independent experiments. The data were analyzed using Kruskal-Wallis test.
|
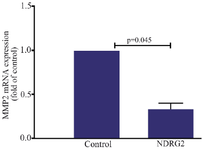
Figure 5. NDRG2 overexpression reduced MMP-2 expression in the A549 cell line. The data represent the mean±SD of at least three independent experiments. The data were analyzed using Mann-Whitney test.
*p<0.05 was considered statistically significant
|

Table 1. The primer sequences for the real-time PCR analysis of NDRG2 and MMP-2 gene expressions
|
|