Development of a Sensitive Enzyme-Linked Immunosorbent Assay for Detection of Hepatitis B Surface Antigen Using Novel Monoclonal Antibodies
-
Yazdani, Yaghoub
-
Department of Immunology, School of Public Health, Tehran University of Medical Sciences , Tehran, Iran
-
Department of Molecular Medicine, Faculty of Advanced Medical Science Technologies, Golestan University of Medical Sciences , Gorgan, Iran
-
Roohi, Azam
-
Monoclonal Antibody Research Center, Avicenna Research Institute, ACECR , Tehran, Iran
-
Khoshnoodi, Jalal
-
Department of Immunology, School of Public Health, Tehran University of Medical Sciences , Tehran, Iran
-
 Shokri, Fazel
Ph.D., Department of Immunology, School of Public Health, Tehran University of Medical Sciences, Tehran, Iran, Tel: + 98-21-88953021 E-mail: fshokri@sina.tums.ac.ir
Shokri, Fazel
Ph.D., Department of Immunology, School of Public Health, Tehran University of Medical Sciences, Tehran, Iran, Tel: + 98-21-88953021 E-mail: fshokri@sina.tums.ac.ir
-
Department of Immunology, School of Public Health, Tehran University of Medical Sciences , Tehran, Iran
-
Monoclonal Antibody Research Center, Avicenna Research Institute, ACECR , Tehran, Iran
Abstract: Hepatitis B virus (HBV) infection is the 10th leading cause of death worldwide. The most important diagnostic and screening marker for HBV infection is Hepatitis B surface antigen (HBsAg), and the most widely used HBsAg screening test is Enzyme-linked Immunosorbent Assay (ELISA). In this study, an ELISA assay has been developed for detection of HBsAg using two novel monoclonal antibodies (mAb) as capture layer and a polyclonal biotinylated Ab as detector phase. We evaluated the sensitivity, specificity, detection limit, seroconversion time, positive and negative predictive values and reproducibility of our assay with standard panels and different serum samples. The results were compared with a well established commercial kit. Both assays showed similar detection limit values of 0.5 to 0.7 ng/ml and the same seroconversion periods of 42 and 65 days for “ad” and “ay” serotypes of HBsAg, respectively. Sensitivity and specificity of the assay were 98.98% and 99.6%, respectively. The positive and negative predictive values of our assay were also calculated as 99.49% and 99.2%, respectively. Analysis of reproducibility of the present assay demonstrated 3.96% and 5.85% intra-and inter-assay coefficient of variations, respectively, which were less than those obtained by the commercial kit. There was a highly significant correlation between our designed assay and the commercial ELISA kit (p < 0.0001, r = 0.957). Altogether, our results indicate that the designed assay is comparable to the commercial kit in terms of sensitivity, specificity, positive and negative predictive values and reproducibility and could be employed for diagnosis of HBV infection in blood samples.
Introduction :
Hepatitis B virus (HBV) infection is a global health issue and it is estimated that approximately 360 million people are chronic carriers of the HBV (1, 2). HBV has a double stranded DNA encoding for P, X, core and surface proteins (3). In the course of infection, an array of these proteins may be detected in the blood. Amongst these proteins, Hepatitis B surface antigen (HBsAg) is the earliest indicator of infection which may identify infected people before appearance of clinical symptoms (4). During the recovery period, HBsAg disappears from the blood; however, this protein remains positive in chronic carriers (5, 6). Hence, HBsAg detection is the most important approach to determine chronic and acute HBV infection.
Antibodies against HBV proteins are other immunological markers of infection, of which anti hepatitis B core antigen (anti-HBc) appears shortly after HBsAg and is an important marker of past or present HBV infection (7).
Both serological and molecular screening tests are employed for the diagnosis and monitoring of HBV infection (8). Although molecular Nucleic Acid Testing (NAT) is preferable in terms of its simplicity, rapidness and sensitivity (9, 10), yet for blood screening and diagnosis, especially in some cases of occult HBV (11, 12) that NAT may miss some positive samples, there is a room to improve serological assays (13). The ability of NAT to decrease window period is controversial (14, 15). It is also shown that NAT sensitivity is affected by viral load and pool size and it is too costly to test individual samples by NAT (16). Even by using individual HBV NAT, serological screening may be necessary to prevent some HBV transmission through blood transfusion (9, 16).
Among all HBsAg assays, enzyme-linked immunosorbent assays (ELISA) are most frequently used because of their sensitivity and cost-effectiveness (17). In the blood banks of all developed countries screening for hepatitis B virus (HBV) using enzyme immunoassay (EIA) is mandatory (18).
Recent studies have revealed that quantitative monitoring of serum HBsAg may be used as a novel tool for the assessment of antiviral therapy efficacy, because there is a correlation between concentration of HBsAg and HBV- DNA (19-21). Seroconversion period is one of the most important features of the HBsAg assays. This period has been estimated to be 35 days by individual sample NAT, 41 days by minipool NAT, and 50 days by highly sensitive HBsAg immunoassays (22).
The other important characteristic of the ELISA assays which needs continuous improvement is their capability to detect mutant HBV isolates by employment of new monoclonal antibodies (mAbs) specific for HBsAg (22). In the present study, a sensitive ELISA was developed by incorporating two novel mAbs which have recently been shown to detect a variety of mutant HBsAgs from all major subtypes (23).
Materials and Methods :
Antibody production and conjugation
Nine murine monoclonal antibodies (mAbs) and rabbit polyclonal antibodies against recombinant adw subtype of HBsAg (Heberbiovac, Cuba) were produced, purified, characterized, conjugated with Horseradish Peroxidase (HRP) and biotinylated as previously described (23, 24).
Reactivity pattern of mAbs with three different HBsAg subtypes
To study interaction between ayw, adr and adw subtypes of HBsAg with mAbs raised against adw subtype, an indirect ELISA was performed. Polystyrene plates (Maxisorp, Nunc, Denmark) were coated in triplicate with 1 µg/ml of rHBsAg diluted in phosphate buffered saline (PBS, 0.15 M, pH=7.2) for 90 min at 37 oC. Three wells were coated with 1µg/ml of Bovine Serum Albumin (BSA, Sigma, USA) as negative control. After washing with PBS containing 0.05% Tween 20 (Sigma, USA) and blocking with PBS containing 3% skim milk, 10 µg/ml of mAbs were added to the plate followed by washing steps. Appropriate dilution of Horseradish Peroxidase (HRP)-conjugated sheep anti-mouse Ig (prepared in our lab) was then added and reaction revealed with O-phenylendiamine dihydrocloride (OPD, Sigma) as substrate. HRP-conjugated sheep anti mouse was added to wells coated with rHBsAg (without mAbs) to determine background reaction. Finally, the reaction was stopped with 20% H2SO4 and Optical Density (OD) measured by a multiscan ELISA reader (Organon Teknika, Boxtel, The Netherlands) at 492 nm.
Design of ELISA for determination of HBsAg
Polystyrene microtiter plates were coated in triplicate with 100 µl of 10 µg/ml of unlabelled mAbs diluted in PBS for 90 min at 37 oC. Three wells were coated with 3% skim milk as negative control. After washing with PBS and blocking with PBS containing 3% skim milk, different dilutions of purified rHBsAg or serum samples were added to the plates followed by washing steps. Appropriate dilutions of biotinylated anti-HBsAg mAbs or polyclonal antibodies were then added. After washing, appropriate dilution of HRP-conjugated Strepavidin (Sigma, USA) was subsequently added and the reaction revealed using 3, 3', 5, 5'-Tetramethyl Benzidine (TMB, Sigma, USA) as substrate. HRP-conjugated Strepavidin was added to wells coated with unlabelled mAbs (without rHBsAg or serum samples) to account for background reactivity. Finally, the reaction was stopped with 20% H2SO4 and OD measured at 450 nm.
Determination of the detection limit of the assay
Detection limit of the designed assay was evaluated using decreasing concentrations of three serotypes of purified HBsAg (“ayw”, “adr” and “adw”). A standard sensitivity panel (PHA 807, BBI Co, USA) was also employed to determine sensitivity of our designed ELISA assay. Two standard seroconversion panels (PHM 912 and PHM 916, BBI Co, USA) were used to determine the earliest seroconversion time which could be identified by this assay. Samples included in the three standard panels were undiluted sera collected at different time points from subjects infected with “ay” or “ad” HBs serotype at early stage of infection.
Assay validation and statistical analysis
Specificity, sensitivity, positive predictive value and negative predictitive value of the designed ELISA assay were determined using three collections of human sera including 198 HBsAg positive, 250 HBsAg negative and 100 unselected samples collected from the Iranian Blood Transfusion Organization and referral medical labs, respectively. Seropositivity for HBsAg was approved by a commercial ELISA kit employed as a gold standard (Hepanostika; bioMerieux, France). Intra- and inter-assay variations were determined using two pooled HBsAg positive sera. Then, Intra- and inter-assay variations were calculated using the formula:
where CV= coefficient of variation, SD=standard deviation and X=mean. Correlation between the results of the designed assay and the commercial kit was determined by Spearman's correlation test and values of less than 0.05 were considered significan
Result :
Reactivity pattern of HBsAg –specific mAbs with HBsAg serotypes
Nine HBsAg-specific mAbs were selected and evaluated in this study. Specificity and immunochemical characteristics of the selected mAbs have previously been described (23, 24). The results of reactivity pattern of these mAbs with “adw”, “ayw” and “adr” serotypes are summarized in table 1.
Selection of capture and detection mAbs
To identify the best combinations of mAbs to be used as capture and detector pair in our designed ELISA assay, we checked mAbs which had high reactivity to rHBsAg as solid phase (unlabelled capture) and liquid phase (biotynilated detector) Abs pairwise (data not presented). Based on our results, “34D7/S3” and “33D7/S4” were selected as suitable candidates for detection and “37C5/S7” and “35C6/S11” as capture mAbs.
Reactivity of the two selected detector mAbs was compared with purified polyclonal anti-HBsAg antibodies (pAbs). Appropriate concentrations of biotynilated “34D7/S3” and “33D7/S4” mAbs were mixed and compared with pre-selected concentration of the biotynilated pAb. As illustrated in figure 1, biotynilated anti-HBsAg pAb displayed substantially better reactivity than the combination of the two selected mAbs. Thus, biotynilated pAb was subsequently employed as detector probe in our designed ELISA assay.
Detection limit of the designed ELISA assay
Using this assay, down to 0.5 ng/ml of HBsAg was detectable (Figure 2). Using the standard BBI sensitivity panel, this value was 0.7 and 0.5 ng/ml for “ad” and “ay” subtypes of HBsAg, respectively (Table 2). These results are comparable to the commercial ELISA kit employed in this study.
Determination of seroconversion time
Two BBI standard seroconversion panels were used to determine the earliest seroconversion time identifiable by the designed assay. Infection with “ad” and “ay” serotypes of HBV could be identified as early as 42 and 65 days post infection, respectively (Table 3). Similar results were obtained by the commercial kit.
Specificity and sensitivity of the assay
Specificity and sensitivity of the designed assay were determined using two panels of positive (n=198) and negative (n=250) serum samples and a panel of unselected samples (n=100). The results are presented in table 4. Based on these results, sensitivity and specificity of our designed assay was found to be 98.98% and 99.6%, respectively. The positive and negative predictive values of the assay were also calculated as 99.49% and 99.2%, respectively. A highly significant correlation was observed between the designed assay and the commercial kit (p<0.0001) (Figure 3).
Assay reproducibility
Assay variation was checked using two serum samples with different concentrations of HBsAg. Each sample was run eight times to calculate the Coefficient of Variation (CV). Intra-assay and inter-assay CV were found to be 3.96% and 5.85% for the designed assay and 4.5% and 9.29% for the commercial kit, respectively.
Discussion :
Improvement of immunoassays for detection of viral infections, particularly HBV infection, has always been considered as a health priority, due to emergence of mutant isolates and the need to early diagnosis of the disease. Both requirements are achieved by employment of new mAbs with improved affinity and specificity. In the present study, novel HBsAg-specific mAbs were employed to develop a sensitive sandwich ELISA for detection of HBsAg. Different combinations of the mAb which had high reactivity with rHBsAg (Table 1) were applied to select the best capture and detector pairs. Our results showed that “37C5/S7” and “35C6/S11” were the most active capture mAbs and “34D7/S3” and “33D7/S4” were the best detector mAbs (data not presented). Interestingly, the two capture mAbs have already been shown to recognize linear epitopes within the "a" region of HBsAg, making them more suitable targets for detection due to their stability (23, 24).
Acknowledgement :
This study was supported in part by a grant from Tehran University of Medical Sciences
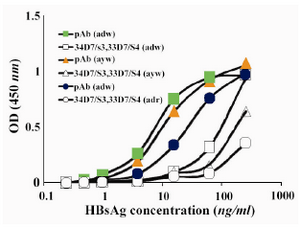
Figure 1. Comparison between reactivity of a combin-ation of the two selected mAbs and polyclonal antibody as detector phase in the designed ELISA assay pAb: polyclonal antibody; mAb: monoclonal antibodies (a combination of 34D7/S3 and 33D7/S4 mAbs)
|
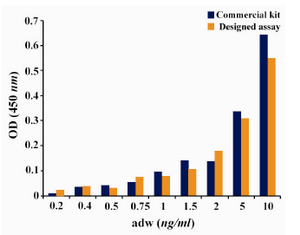
Figure 2. Comparison of detection limit of the designed assay and commercial kit
|
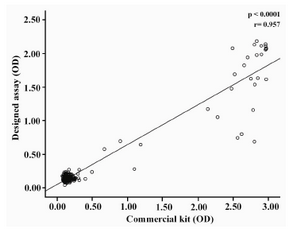
Figure 3. Correlation between the results obtained by the designed ELISA assay and commercial kit
|
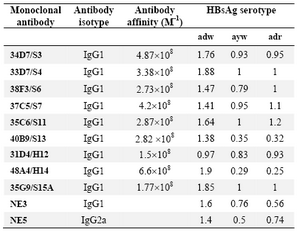
Table 1. Reactivity pattern of the selected mAbs with different HBsAg serotypes
The results represent OD values.
NE3 and NE5 are commercial anti-HBs mAbs recognizing the "a" determinant (IbtGMbh aspenhouster,Germany) which have been employed as control
|
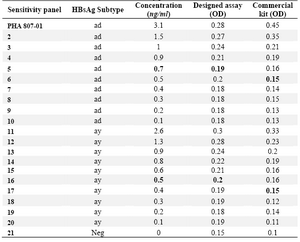
Table 2. Determination of detection limit of the designed ELISA assay using the standard BBI sensitivity panel
Samples with OD mean of triplicate Neg (sample NO.21)+0.05 were considered reactive
|
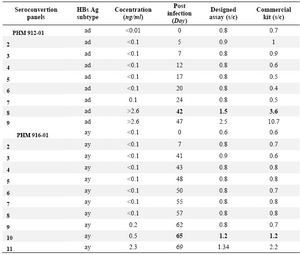
Table 3. Determination of the seroconvertion time of HBV infection by the designed ELISA assay using two BBI standard seroconvertion panels
Samples with signal-to cutoff (s/c)>1.0 were considered reactive
|
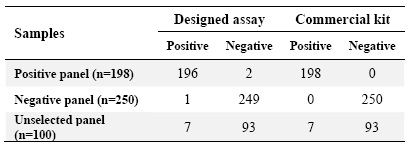
Table 4. Reactivity of the designed ELISA assay with different panels of serum samples
Unselected panel refers to serum samples collected from different referral hepatitis diagnostic laboratories without prior information about their hepatitis B status. Positive and negative panels were collected from the Iranian Blood Transfusion Organization based on reactivity with the commercial ELISA kit which was employed as a gold standard
|
|