The Impact of Nrf2 Silencing on Nrf2-PD-L1 Axis to Overcome Oxaliplatin Resistance as well as Migration in Colon Cancer
-
Payandeh, Zahra
-
Immunology Research Center, Tabriz University of Medical Sciences, Tabriz, Iran
-
Pirpour Tazehkand, Abbas
-
Department of Biochemistry and Clinical Laboratories, Faculty of Medicine, Tabriz University of Medical Sciences, Tabriz, Iran, Tabriz, Iran
-
Mansoori, Behzad
-
Immunology Research Center, Tabriz University of Medical Sciences, Tabriz, Iran
-
Department of Cancer and Inflammation Research, Institute for Molecular Medicine, University of Southern Denmark , Odense, Denmark
-
Drug Applied Research Center, Tabriz University of Medical Sciences, Tabriz, Iran
-
Khaze, Vahid
-
Immunology Research Center, Tabriz University of Medical Sciences, Tabriz, Iran
-
Asadi, Milad
-
Immunology Research Center, Tabriz University of Medical Sciences, Tabriz, Iran
-
 Baradaran, Behzad
Immunology Research Center, Tabriz University of Medical Sciences, Tabriz, Iran, Tel:+98 9391559561, 4133371440; Email: baradaranb@tbzmed.ac.ir
Baradaran, Behzad
Immunology Research Center, Tabriz University of Medical Sciences, Tabriz, Iran, Tel:+98 9391559561, 4133371440; Email: baradaranb@tbzmed.ac.ir
-
 Samadi , Nasser
Department of Biochemistry and Clinical Laboratories, Faculty of Medicine, Tabriz University of Medical Sciences, Tabriz, Iran, Tel: +98 9391559561, 4133371440; Email: nsamadi@ualberta.ca
Samadi , Nasser
Department of Biochemistry and Clinical Laboratories, Faculty of Medicine, Tabriz University of Medical Sciences, Tabriz, Iran, Tel: +98 9391559561, 4133371440; Email: nsamadi@ualberta.ca
Abstract: Background: Nuclear factor-erythroid 2-related factor 2 (Nrf2) plays a key role in promoting chemoresistance in various cancers. PD-L1 is one of the downstream targets of the Nrf2 signaling pathway. This molecule has some beneficial impacts on tumors growth by inhibition of the immune system. This study aimed to investigate the potential role of the Nrf2-PD-L1 axis in the promotion of oxaliplatin resistance in colon cancer cells.
Methods: We examined Nrf2, PD- L1, and CD80 expression in the tumor and margin tissue samples from CRC patients. After that role of the Nrf2-PD-L1 axis in promotion of Oxaliplatin resistance was investigated.
Results: Our data revealed that Nrf2 and PD-L1 mRNA expressions were markedly higher in tumor tissues compared to margin tissues. The PD-L1 mRNA expression level was also increased in the resistant cells. However, Nrf2 expression was decreased in SW480/Res cells and increased in LS174T/Res cells. The inhibition of Nrf2 through siRNA treatment in SW480/Res and LS174T/Res cells has decreased the IC50 values of oxaliplatin. Inhibition of the Nrf2 has significantly increased the oxaliplatin-induced apoptosis, and reduced the migration in SW480/Res cells.
Conclusion: It is suggested that effective inhibition of Nrf2-PD-L1 signaling pathways can be considered as a novel approach to improve oxaliplatin efficacy in colon cancer patients.
Introduction :
Colorectal Cancer (CRC) is one of the most prevalent cancers worldwide, which is routinely diagnosed at a higher rate of distant metastases 1. Oxaliplatin resistance is a common phenomenon in the failure of treatment following the surgery 2. Nuclear factor−erythroid 2−related factor 2 (Nrf2) plays a key role in the protection of cells against oxidative stress and inflammation in numerous tissues 3 and its role has been investigated in cancer stem cells 4,5.
Nrf2 overexpression has been reported in diverse types of tumors, associated with disease-free and shorter overall survival 6-8. Programmed Death-Ligand 1 (PD-L1) molecule is classified as a member of the CD28-B7 family which is commonly expressed on the surface of tumor cells 9,10. Having promising clinical outcomes in different types of cancers, various antibodies against PD-1 or PD-L1 have recently gained massive attention 11-13. However, resistance to these antibody-based treatments has been reported. To circumvent this drawback, combined therapies for advanced melanoma are tested. Up to now, several upstream regulators of PD-L1 have been announced.
Identifying and developing alternative therapies for inhibition of the PD-1/PDL1 is necessary to overcome drug resistance in cancer patients. Yin et al have conducted a study to show that Nrf2 is the upstream regulator of the PD-L1. Destruction or suppression of Nrf2 has inhibited the progression of melanoma through a significant increase in the activity of both CD4+ and CD8+ cells; Nrf2 inhibition in combination with anti-PD-1 therapy has increased the anti-tumor efficacy 14. Altogether, all of these studies clarified that Nrf2 suppression is a beneficial strategy in cancer treatment 15,16.
The present study aimed to investigate the potential role of Nrf2, PD- L1, and CD80 in the promotion of oxaliplatin resistance in colon cancer cells. In this regard, their expression was examined in the tumor and margin of tissue samples from CRC patients. Then, the potential role of the Nrf2-PD-L1 axis in promotion of oxaliplatin resistance (In colon cancer cells) was investigated. The obtained results revealed that the effective inhibition of the Nrf2-PD-L1 axis can be considered as a promising approach for the treatment of colon cancer patients.
Materials and Methods :
Patients and specimens: This study was conducted under the institutional bioethical guidelines of the Ethics Committee at Tabriz University of Medical Sciences (IR.TBZMED.VCR. REC.1397.403). Tumor and matched marginal tissues (N=50) were collected during surgery from patients who referred to Imam Reza Hospital at Tabriz University of Medical Sciences. Written informed consent was obtained from all patients. All tissue samples were immediately transferred into RNAase inhibitor solution (Qiagen, Germany) and stored at -80°C for further experiments.
Cell culture and treatment: SW480 and LS174T colon cancer cells were purchased from the National Cell Bank of Iran (Pasteur Institute, Iran). SW480 and LS174T resistant cells were generated as previously described 17-19. SW480 and SW480 resistant cells were cultured in RPMI-1640 medium (Gibco, Thermo Fisher Scientific, USA); LS174T and LS174T resistant cells were cultured in DMEM high glucose medium (Gibco, Thermo Fisher Scientific, USA) which all were supplemented with FBS 10% (Gibco, commercial cat. no. 10437028, Thermo Fisher Scientific, USA). The cells were incubated in a humidified atmosphere containing 5.2% CO2 at 37°C. The sensitive and resistant SW480 colon cancer cells were treated with Brusatol (40 nM) and Luteolin (20 µM) alone or in combination and tert-buthylhydroquinone (tBHQ) (20 µM) for 24 hr.
Gene expression analysis through quantitative real-time PCR: Total RNA was extracted via RiboEx reagent (GeneAll, Korea) according to the manufacturer’s protocol. The total RNA of clinical tissue was extracted via grinding them in 1 ml RiboEx reagent using porcelain glass grinder. Complete DNA (cDNA) was synthesized from 1 µg of total RNA using cDNA synthesis kit (Biofact, Korea). Then, qRT-PCR technique was applied to assess gene expressions. First, total RNA of tumor tissues and cells were extracted using the RiboEx reagent (GeneAll, Korea) according to the manufacturer's protocol. Then, total RNA was quantified and qualified by NanoDrop Spectrophotometer (Thermo Fisher Scientific, USA) and agarose gel electrophoresis, respectively. Thereafter, cDNA was synthesized by a cDNA synthesis kit (Takara, Japan). Finally, mRNA expressions were examined by the Roche LightCycler system (Roche, Germany) by applying SYBR Premix Ex Taq (Takara, Japan). Relative Nrf2, PD-L1, and CD80 mRNA expressions in tissue samples were analyzed through the 2−ΔΔCt method, in which endogenous β actin was considered as an internal control. However, for the assessment of raw data from the cell samples, Pffafl method was applied and GAPDH gene was considered as an internal control. All of the experiments were performed at least in triplicate.
siRNA transfection: First, SW480, SW480/Res and LS174T, and LS174T/Res cells were seeded with a density of 5×105 cells per well in a six-well plate and allowed to reach 60 to 70% confluency. Then, culture media were removed and siRNA transfection (Santa Cruz Biotechnology, USA) was performed by applying transfection reagent according to the manufacturer’s instruction. Briefly, siRNA (80 pmol) and transfection reagent (8 pmol) were separately diluted in the siRNA transfection medium and incubated at room temperature for 10 min. Then, they were mixed and incubated at room temperature for 20 min or more. Finally, the mixture was transferred to the wells comprising cells and transfection medium; the cells were incubated about 6 hr at 37°C in a CO2 incubator. Next, the cells were cultured in the RPMI‐1640 medium containing 20% FBS. After 48 hr, the cells were used for further experiments.
Assessment of cell viability through MTT assay: First, the cells were seeded in the 96-well plates with a density of 104 cells/well in triplicate. Then, the cells were incubated for 24 hr or overnight. After removing the media, the cells were incubated with different oxaliplatin concentrations (1 μM to 100 μM) in the presence and absence of Nrf2 inhibitor (Nrf2 siRNA). After 24 hr, the medium was removed and replaced with fresh medium containing 3-(4, 5- dimethyl-
thiazol-2-yl)-2, 5-diphenyl tetrazolium bromide (MTT) (2 mg/ml) and cells were further incubated for 4 hr at 37°C. Then, the medium was replaced with 200 μl DMSO following gentle shaking for 15 min. Finally, Sunrise microplate reader (Tecan, Australia) was used to measure the optical density (OD) of the wells at a wavelength of 570 nm 20; IC50 of drugs on the cells was calculated using GraphPad prism v.7 through non-linear regression analysis.
Apoptosis assay: Annexin V/PI assay was applied to examine the impact of Nrf2 siRNA on the oxaliplatin-induced apoptosis in SW480/Res cells. First, SW480/Res cells were seeded in a six-well plate (2×105 cells per well). Then, the cells were transfected with Nrf2 siRNA and treated with oxaliplatin for 48 hr. The cells were harvested using trypsin EDTA 0.05%. Subsequently, the cells were washed twice with PBS, and stained by applying ApoFlowEx® FITC Kit (EXBIO Praha, a.s., Czech Republic). Finally, the apoptotic cells were analyzed through MACSQuant® 10 Flow Cytometer (Miltenyi Biotec, Germany). FlowJo (7.6.1) software was used for analysis of raw data 21,22.
Wound-healing assay: Wound-healing assay was applied to investigate the impact of Nrf2 siRNA on the SW480/Res cell migration capacity. The cells with a density of 50,000 cells per well were seeded into a 24‐well plate and incubated to reach the 90% confluency or more in the monolayer. Then, to create a "wound", the layer was scratched by applying a sterile yellow micropipette tip and the cell debris was washed with serum-free media. After Nrf2 siRNA transfection, the wound images were captured using the light microscope at different times (0, 6, 12, 24, and 48 hr post scratching). The migration capacity was calculated by measuring the distance between the wound edges through Image J software. The wound-healing assay was performed in triplicate.
Statistical analysis: All experiments were performed in triplicate or more. Data analysis was performed using one-way ANOVA or unpaired t-test using GraphPad prism (v 10.00) software. The p<0.05 was considered statistically significant.
Results :
Nrf2, PD-L1, and CD80 gene expression levels in colon cancer tissues: qRT-PCR was applied to examine the Nrf2, PD-L1, and CD80 gene expression levels in CRC tissues. Our results showed that Nrf2 and PD-L1 expressions were markedly higher in tumor tissues compared to margin tissues (p<0.001). However, the difference in CD80 expression was not statistically significant among the groups (p<0.001) (Figure 1). Moreover, the correlation between Nrf2, PD-L1, and CD80 expressions was determined through Spearman's rank correlation. Our results revealed a positive correlation between Nrf2 and PD-L1 gene expressions in CRC samples (r= 0.8434, p<0.001).
The impact of Nrf2 inhibition on PD-L1 expression: SW480 and LS174T sensitive and resistant cells were treated with Brusatol (40 nM) and Luteolin (20 µM) alone and in combination for 24 hr (Chemical inhibitors of Nrf2). Nrf2 inhibition by Brusatol, Luteolin, and their combination in SW480 cells decreased Nrf2 mRNA level about five, four and five-fold, and PD-L1 expression ten, seven and ten, respectively. However, Nrf2 and PD-L1 expressions were increased about three-fold after applying tBHQ as Nrf2 activator (Figure 2; up: A-B). In addition, our results from SW480/Res cells showed that Nrf2 mRNA levels were respectively decreased about thirteen, five and twelve-fold after inhibition by Brusatol, Luteolin, and their combination. However, Nrf2 expression was increased about five-fold after applying tBHQ. In SW480/Res cells, Nrf2 inhibition by Brusatol, Luteolin, and their combination respectively caused five, four, and five-fold decreases in PD-L1 mRNA levels; PD-L1 expression was increased about three-fold in the tBHQ treated groups (Figure 2; up: C-D). Brusatol, Luteolin, and their combination in LS174T cells decreased Nrf2 mRNA level about six, eleven and fourteen-fold, respectively, and also decreased PD-L1 expression about five, eight, and five, respectively. However, Nrf2 expression was increased about three-fold after its activation through tBHQ, increasing PD-L1 expression about four-fold (Figure 2; down: A-B). Furthermore, in LS174T/Res cells, Nrf2 inhibition by Brusatol, Luteolin, and their combination caused eleven, ten and eleven-fold decreases in Nrf2 mRNA level, respectively. However, tBHQ increased Nrf2 expression about three-fold in the cells. Nrf2 inhibition by Brusatol, Luteolin alone, and in combination caused nine, eight and ten-fold decreases in PD-L1 mRNA level. But PD-L1 expression was increased about three-fold in the tBHQ treated groups (Figure 2; down: D).
Nrf2 silencing and downregulation in Nrf2 and PD-L1: To investigate the impact of Nrf2 silencing on Nrf2 and PD-L1 expressions, SW480, SW480/Res, LS174T, and LS174T/Res colon cancer cells were transfected with 80 pmol of Nrf2 siRNA for 48 hr. Then, the gene expressions were evaluated through qRT-PCR. Our results showed that by Nrf2 silencing, Nrf2 and PD-L1 expressions were downregulated about 51, 55, 36 and 49.5% in the SW480, SW480/Res, LS174T and LS174T/Res cells, respectively (p<0.05) (Figure 3; up: A). In addition, with this transfection, PD-L1 expression was decreased around 51, 55, 36, and 49.5% in the SW480, SW480/Res, LS174T and LS174T/Res cells, respectively (p<0.05) (Figure 3; up: B).
To evaluate the impact of Nrf2 inhibition on oxaliplatin efficacy to overcome oxaliplatin resistance in colon cancer cells, two oxaliplatin resistant colon cancer cells (SW480/Res and LS174T/Res cells) were applied. The cells were transformed with Nrf2 siRNA or NC siRNA and treated with different oxaliplatin concentrations for 48 hr. Then, the IC50 values of the oxaliplatin and Nrf2 siRNA alone and in combination were determined using MTT assay. Our results showed that IC50 values of oxaliplatin in SW480 and LS174T cells increased from 10.56 and 8.83 μM to 89.60 and 43.34 in resistant cells, respectively. Nrf2 inhibition by siRNA in SW480/Res and LS174T/Res decreased the IC50 values of oxaliplatin from 89.60 and 43.34 μM to 23.08 and 12/25 μM, respectively (p<0.05). Furthermore, IC50 values of Nrf2 siRNA were determined to be 34.22 and 10.29 μM for SW480/Res and LS174T/ Res cells, respectively (Figure 3; down).
The impact of Nrf2 silencing on oxaliplatin-induced apoptosis and cell migration in SW480/Res cells: To investigate the impact of Nrf2 silencing on the oxaliplatin-induced apoptosis, annexin V/PI was applied. SW480/Res cells were treated with oxaliplatin (90 μM) and Nrf2 siRNA (80 pml) alone and in combination for 48 hr. The cells were stained with annexin V/PI and apoptotic cells were detected by flow cytometry. Our data showed that the apoptosis rates were 40.18% (35.6%+5.13%) for oxaliplatin, 4.47% (2.74%+1.73%) for Nrf2 siRNA, and 40.22% (32.4+ 8.18) for combination of Nrf2 siRNA and oxaliplatin treated groups (Figure 4A). A wound-healing assay was used to investigate the impact of Nrf2 silencing on migration in SW480/Res cells. First, the cells’ monolayer was scratched and further treated with oxaliplatin (90 μM) and Nrf2 siRNA (80 pml) alone and in combination. Our findings showed that Nrf2 knocking down reduced migration of the cells in the scratched area, which was more remarkable in the presence of oxaliplatin (Figure 4B).
Discussion :
Despite the advances in the treatment of colon cancer in the last decades, the growing death rate is still statistically remarkable in the patients 23. It was demonstrated that NRF2 is an upstream transcriptional activator of PDL1 which regulates its expression 14. Also, Nrf2 and PD-L1 signaling pathways were reported to have co-regulatory functions in melanoma. It has also been shown that Nrf2 regulates the expression of PD-L1, which can be considered as a potential alternative strategy for PD-1/PD-L1 antibody-based treatment of melanoma 14. To the best of our knowledge, there are no studies to investigate the relationship between the Nrf2-PD-L1 axes in promoting oxaliplatin resistance in colon cancer.
Therefore, the importance of the Nrf2-PD-L1 axis in resistance and migration of colon cancer cells was clarified in this study. Primarily, the expression of Nrf2, PD-L1, and CD80 genes was examined in CRC biopsy specimens. Our results showed that Nrf2 was significantly overexpressed in CRC samples compared to the margin tissues. In concordance with our study, several studies on tissue samples taken during colonoscopy revealed that Nrf2 is overexpressed in the patients 24. Besides, Nrf2 overexpression was in concordance with the stage and grade of tumors 25.
It has also been shown that Nrf2 overexpression has a vital function in the pathogenesis of CRC. Then, PD-L1 mRNA expression was investigated in CRC, showing that PD-L1 was up-regulated in CRC tissues versus to margin tissues. Also, a positive correlation was found between Nrf2 and PD-L1 mRNA expressions in tumor tissues. Consistently, Nrf2 positive control on PD-L1 expression has been shown 14.
Chemical inhibition of Nrf2 with Brusatol and Luteolin showed a strong reduction of Nrf2 and PD-L1 mRNA expression levels. This result supports the idea that Nrf2 positively controls the PD-L1 expression. Consistently, Nrf2 activation by tBHQ has also strongly increased Nrf2 and PD-L1 mRNA expression levels. Sabzichi et al showed that Nrf2 activation by 20 μM of tBHQ has increased Nrf2 mRNA expressions 26. Moreover, the impact of Nrf2 inhibition on PD-L1 expression was studied by applying Nrf2 siRNA. The siRNAs are the powerful tools to silence target genes in mammalian cells.
Our results revealed that Nrf2 silencing has significantly decreased Nrf2 and PD-L1mRNA expressions in the sensitive and resistant cells in a time-dependent manner. Moreover, Nrf2 silencing was applied to evaluate its potential impacts on oxaliplatin efficacy to break down oxaliplatin resistance in SW480/Res, and LS174T/Res cells. The results from MTT and apoptosis assays showed a strong synergistic effect of Nrf2 knocking down with oxaliplatin in both SW480/Res and LS174T/Res cells. Previously, it was demonstrated that Nrf2 inhibition by Brusatol promotes the oxaliplatin−induced cytotoxicity and apoptosis in the cells 27. It has been demonstrated that a recombinant NRF2-siRNA significantly downregulates the NRF2-dependent ATP-Binding Cassette (ABC) efflux transporters, and increases the sensitivity of MG63 and 143B cells to sorafenib, doxorubicin, and cisplatin 28.
Conclusion :
In this study, the potential role of Nrf2 siRNA was examined in inhibition of migration in SW480/Res colon cancer cells through wound-healing (Scratch) assay. The results showed that treatment of SW480/Res cells with Nrf2 siRNA and oxaliplatin alone or in combination significantly inhibited migration. The maximum inhibition was observed when the cells were treated with a combination of Nrf2 siRNA and oxaliplatin. In concordance with our results, it has been shown that Nrf2 inhibition by chrysin suppresses the proliferation, invasion, and migration in glioblastoma cells 29. Furthermore, it has been shown that silencing of the Nrf2 gene promotes the efficacy of chemotherapy and suppresses tumor growth in non-small cell lung cancer 30.
Acknowledgement :
The authors wish to thank the staff at Immunology Research Center of Tabriz University of Medical Sciences, Iran
Conflict of Interest :
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Funding: This work was financially supported by grants from the Iran National Science Foundation (INSF), Tehran, Iran (Project no. 97021150) and Tabriz University of Medical Sciences, Tabriz, Iran (Project no. 61892). We gratefully acknowledge their contribution to this study.
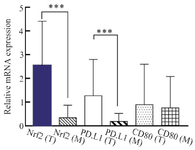
Figure 1. The mRNA expression level of Nrf2, PD-L1, and CD80 in the tumor (T) and margin (M) tissues of colon cancer patients. Nrf2 and PD-L1 were significantly overexpressed in tumor tissues in comparison to margin ones (p<0.001). There were not any significant differences in CD80 mRNA expression between tumor and margin tissues. The results were gathered at least from three independent experiments and finally expressed as mean±SD. *** p<0.001.
|
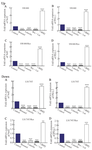
Figure 2. Up) the impact of Nrf2 inhibition by Brusatol (Bru) and Luteolin (Lut) and activation by tBHQ on Nrf2 and PD-L1 mRNA expression levels. The sensitive and resistant (Res) SW480 colon cancer cells were treated with Brusatol (40 nM) and Luteolin (20 µM) alone or in combination and tert-buthylhydroquinone (20 µM) for 24 hr. The results were gathered at least from three independent experiments and finally expressed as mean±SD. *** p<0.001. Down) the impact of Nrf2 inhibition by Brusatol (Bru) and Luteolin (Lut) and activation by tBHQ on Nrf2 and PD-L1 mRNA expression levels. The sensitive and resistant (Res) LS174T colon cancer cells were treated with Brusatol (40 nM) and Luteolin (20 µM) alone or in combination and tert-buthylhydroquinone (20 µM) for 24 hr. The results were gathered at least from three independent experiments and finally expressed as mean±SD. *** p<0.001.
|
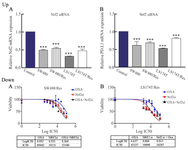
Figure 3. Up) the impact of Nrf2 siRNA on Nrf2 and PD-L1 expressions. Sensitive (SW480 and LS174T) and resistant (SW480/Res and LS174T/Res) colon cancer cells were transfected with Nrf2 siRNA (80 pmol) for 48 hr. The results were gathered at least from three independent experiments and finally expressed as mean±SD. *** p<0.001. Down) the impact of Nrf2 siRNA on oxaliplatin efficacy in resistant colon cancer cells. SW480 and LS174T resistant colon cancer cells were transfected with Nrf2 siRNA (80 pmol) which was also followed by different oxaliplatin concentrations for 48 hr. The impacts of Nrf2 siRNA on oxaliplatin induced cytotoxicity in SW480/Res (A) and of LS174T/Res (B) cells.
|

Figure 4. A) the impact of Nrf2 siRNA on oxaliplatin-induced apoptosis in resistant SW480 colon cancer cells. SW480 resistant colon cancer cells were transfected with Nrf2 siRNA (Nrf2 si) (80 pmol) which was also followed by oxaliplatin (Oxp) (89/60 μM) for 48 hr. B) the impact of Nrf2 siRNA on reduction of migration in resistant SW480 colon cancer cells. SW480 resistant colon cancer cells were transfected with Nrf2 siRNA (Nrf2 si) (80 pmol) which was also followed by oxaliplatin (Oxp) (89/60 μM) for 48 hr. Then, the cells’ motility was monitored by wound-healing assay for 0, 24, and 48 hr. The results were gathered at least from three independent experiments and finally expressed as mean±SD. * p<0.05.
|
|