Mitochondrial Distribution and ATP Content of Vitrified, In vitro Matured Mouse Oocytes
-
Nazmara, Zohreh
-
Department of Anatomy, Faculty of Medical Sciences, Tarbiat Modares University, Tehran, Iran
-
 Salehnia, Mojdeh
Department of Anatomy, Faculty of Medical Sciences, Tarbiat Modares University, Tehran, Iran, Tel: +98 21 82883871; E-mail: salehnim@modares.ac.ir
Salehnia, Mojdeh
Department of Anatomy, Faculty of Medical Sciences, Tarbiat Modares University, Tehran, Iran, Tel: +98 21 82883871; E-mail: salehnim@modares.ac.ir
-
Department of Anatomy, Faculty of Medical Sciences, Tarbiat Modares University, Tehran, Iran
-
Hosseinkhani, Saman
-
Department of Biochemistry, Faculty of Biological Sciences, Tarbiat Modares University, Tehran, Iran
Abstract: Background: The objective of this study was to investigate the effect of vitrification and in vitro maturation on the mitochondrial distribution and ATP content of oocytes. Methods: The oocytes at Germinal Vesicle (GV) and Metaphase II (MII) stages were recovered from 6-8 week old NMRI strain female mice. The oocytes were divided into vitrified and non-vitrified groups. Vitrification was done by the cryotop method using ethylene glycol, dimethylsulfoxide and sucrose as cryoprotectants. The GV oocytes were cultured in maturation medium for 24 hrs. The collected in vitro matured oocytes (IVM-MII) and ovulated metaphase II (OV-MII) oocytes were inseminated with capacitated sperm. The ATP content of the oocytes was measured by luciferin-luciferase reaction. Distribution of oocyte mitochondria was studied using Mito Tracker Green staining under fluorescent microscope. Results: The survival rates of vitrified oocytes at GV and MII stages were 87.39 and 89.5%, respectively. There was no significant difference in the developmental and hatching rates of vitrified and non-vitrified oocytes. The ATP content of GV and MII oocytes derived from in vivo and in vitro condition was not significantly different in vitrified and non-vitrified samples. The pattern of mitochondrial distribution in vitrified and non-vitrified GV and MII oocytes was similar but it was different between MII oocytes collected from fallopian tube and in vitro matured MII oocytes. However, the florescent intensity of mitochondrial staining was different in all the groups in the study. Conclusion: Vitrification did not affect mouse oocyte developmental competence, ATP content at different developmental stages but some alteration was seen in mitochondria distribution of in vitro matured oocytes in comparison to their controls.
Introduction :
Oocyte cryopreservation is a useful alternative for fertility preservation in assisted reproductive technologies 1. More than 1000 children are born using this method 2. Two cryopreservation methods including slow freezing and vitrification have been used to preserve oocytes 3. Vitrification method involves rapid cooling and warming rates with a small volume of cryoprotectant solution without ice crystal formation 4. In spite of many advances in the vitrification procedure, this technique is still considered experimental 2. Some mitochondrial changes at the ultrastructural levels were shown after vitrification of oocytes in different mammalian species 5-9. Nagai et al demonstrated that vitrified mouse MII oocyte showed lower developmental rate and the mitochondria had a diffuse and fragmented appearance 8.
The nuclear and cytoplasmic mammalian oocytes maturation is a complex process 10. Nuclear maturation is demonstrated by extrusion of first polar body while there may be no indication for cytoplasmic maturation 11. According to critical role of mitochondria for energy production in oocytes, it can be considered as an indicator of cytoplasmic maturation 12,13.
Impairment of oocyte mitochondria may occur without any detectable morphological changes during cryopreservation or in vitro maturation. Hence, in this study, mitochondria-specific fluorescent probes (Mito Traker Green) were used to show the distribution of mitochondria within the ooplasm.
To our knowledge, no research has been documented for comparing the effect of oocyte vitrification at different developmental stages on the distribution of mitochondria, and the ATP content. Because the function and distribution of mitochondria are critical for oocyte maturation and fertilization thus it would be important to know the possible influences of vitrification and in vitro maturation procedures on oocyte mitochondria. In the present study, an attempt was made to evaluate firstly the effects of developmental stages of oocytes, secondly the vitrification and warming and finally in vitro culture procedure on the oocyte ATP content and mitochondrial distribution.
Materials and Methods :
Animals: Adult female (6-8 weeks old) and male (8-12 weeks old) NMRI mice were cared for and used according to the guide for the care and use of laboratory animals of Tarbiat Modares university. Mice were housed under a 12 hr light: 12 hr dark regimen at 22-24°C and 40-50% humidity with food and water available ad libitum.
Experimental design: The purpose of this study was two fold. First, the effects of vitrification and warming procedure were studied on the developmental competence, mitochondrial distribution and ATP content of vitrified and non-vitrified mouse GV and MII oocytes.
Next, after in vitro culture of both vitrified and non-vitrified oocytes, their mitochondrial distribution and ATP content of collected oocytes from in vivo and in vitro condition were analyzed and compared.
Reagents and culture media: Unless otherwise indicated, all chemicals were purchased from Sigma Aldrich (Germany).
Preparation of oocyte and sperm: Oocyte: Adult female mice (n=30) were superovulated by Intraperitoneal injection (IP) of 10 IU pregnant mare serum gonadotrophin (PMSG, Folligon; Intervet, Australia). To obtain GV oocytes, females were killed by cervical dislocation 48 hr after PMSG injection and dissected ovaries were placed in global total medium (Life Global, USA). Antral follicles were punctured using needles to release the GV oocytes. Cumulus cells were mechanically removed.
For MII oocyte harvesting, female mice (n=50) were superovulated by IP injection of 10 IU PMSG followed with another injection of 10 IU human chorionic gonadotrophin (hCG, Sereno, Switzerland) 48 hr later. The mice were killed by cervical dislocation 12-16 hr after hCG injection and their oviducts were removed.
The Cumulus-Oocyte-Complex (COC) was released with a needle from the ampullary region of each oviduct and then they were exposed to 0.01% hyaluronidase for 1 min to allow cumulus cells to separate from the oocytes. Denuded MII oocytes were washed several times in global total medium. The collected GV and MII oocytes were considered as vitrified and non-vitrified ones.
Sperm: Sperm was obtained from the adult male mice through several slits in the cauda epididymis in global total medium and capacitated for 1.5 hr at 37°C, 5% CO2 in air.
Vitrification and warming: Oocytes were vitrified by cryotop method 14. The Equilibration Solution (ES) contained 7.5% (v/v) Dimethyl Sulphoxide (DMSO) and 7.5% Ethylene Glycol (EG) in Ham's F10 containing 20% Human Serum Albumin (HSA). The Vitrification Solution (VS) contained 15% (v/v) DMSO and 15% EG and 0.5 M sucrose in Ham's F10 containing 20% HSA.
Three-five oocytes were equilibrated in ES for 10 min and then transferred to VS. Finally, the oocytes were loaded onto the strip end of cryotop with a small volume of VS (<0.1 μl) and then plunged into liquid nitrogen (LN) at least for 24 hr.
For warming, the cap of strip was removed and then the strip of cryotop was directly placed into 1 M sucrose at 37°C for 1 min. Next, it was sequentially transferred to 0.5 M sucrose for 3 min, 0.25 M sucrose for 3 min and was washed in Ham's F10. After these steps, GV oocytes were put into in vitro maturation media and MII in culture media, respectively. After 2 hr of incubation, the survival rate of thawed oocytes was assessed.
In vitro maturation: Oocytes were cultured in 20 μl droplet of global media supplemented daily with 75 mIU/ml rFSH (Sereno, Switzerland) and 10 IU/ml hCG under mineral oil at 37°C in 5% CO2 in air for 24 hr. The extrusion of the first polar body was used as the criterion for maturation of GV-stage oocytes 15.
In Vitro Fertilization and culture : Both collected in vitro matured MII oocytes (IVM-MII) and in vivo MII oocytes (OV-MII) were inseminated with sperm in global medium containing 15 mg/ml BSA. After 4-6 hr, oocytes were washed and cultured in 20 μl droplet of global medium under mineral oil at 37°C in 5% CO2 in air. The development of zygotes to hatching stage was recorded daily up to 5 days.
Staining of mitochondria: Oocytes were stained for mitochondria by Mito Tracker Green (Molecular Probes, Eugene, OR) as described previously by Stojkovic et al. Briefly, the oocytes were incubated in pre-warmed and equilibrated global medium (at 37°C in 5% CO2 in air) containing 0.2 mM Mito Tracker Green for 10 min at 37°C. Then the oocytes were washed several times in global medium and mounted on glass slides and observed under fluorescent microscope at 490 wavelengths 10. Then the photos of individual oocytes in each group were captured and imported into Image J software (National Institutes of Health, Bethesda). The fluorescence intensity of each oocyte was quantified and analyzed by this software.
ATP assay: The ATP content of completely denuded single oocytes was measured using methods described previously 16. Briefly, oocytes of all groups (n=20 per each group) were transferred individually in 20 μl of ultrapure water into micro tubes and stored at -80°C until assay. ATP levels were quantified by measuring the luminescence (Berthold LB 9501 luminometer) generated in an ATP-dependent luciferin-luciferase bioluminescence assay (Bioluminescence Somatic Cell Assay System; Sigma, USA). A six-point standard curve (0-5 pmol) was considered in each series of assay. The ATP content was defined according to the standard curve.
Statistical analysis: All experiments were repeated at least four times. The oocyte maturation, fertilization and developmental rates were analyzed by one-way ANOVA and Tukey's HSD was used as the post hoc test. ATP levels and fluorescence intensity of oocytes were analyzed by independent t-test. Statistical significance was described as p<0.05.
Results :
In vitro maturation, fertilization and developmental rates of the oocytes: Data relating to this section is shown in table 1. The survival rate of vitrified-warmed oocytes at GV and MII stages were 87.39 and 89.50%, respectively and there was no significant difference in this regard.
The maturation rates of GV oocytes in vitrified and non-vitrified groups (which reached to MII stage) were 69.48 and 77.45%, respectively and there was no significant difference between the groups.
The fertilization rates in vitrified and non-vitrified IVM-MII oocytes were 64.36 and 72.72% and these rates in OV-MII oocytes were 66.14 and 71.79%, respectively. The blastocyst hatching rates in vitrified IVM-MII and OV-MII groups were 9.89 and 12.14% and these rates in non-vitrified samples were 9.93 and 13.21%, respectively. There was no significant difference in maturation, fertilization and hatching rates between vitrified and non-vitrified oocytes in both in vitro and in vivo samples.
ATP content of oocytes: The comparison of ATP levels in different groups is shown in figure 1. The ATP contents of GV oocyte in vitrified and non-vitrified groups were 0.2692±0.06216 and 0.2974±0.02588 pmol, respectively. The ATP levels in IVM-MII and OV-MII oocytes in control (non-vitrified) group were 0.2970±0.02542 and 0.2850±0.01454 pmol and concentrations for IVM-MII and OV-MII in vitrified group were 0.3165±0.01020 and 0.3165±0.03804 pmol. This content value was not significantly different between groups.
Distribution of mitochondria: The mitochondrial pattern of vitrified and non-vitrified GV oocytes was similar and mitochondria were diffused throughout the ooplasm of GV oocytes in vitrified (Figure 2A) and non-vitrified (Figure 2B) groups.
The mitochondria distribution in MII oocytes obtained from in vitro matured GV oocytes in both non-vitrified (Figure 3A) and vitrified (Figure 3B) groups was similar and they showed homogenous aggregation pattern throughout the ooplasm (spot distribution).
However, in MII oocytes obtained from in vivo condition in both non-vitrified (Figure 3C) and vitrified (Figure 3D) groups, this pattern was not seen. Their mitochondria had diffused pattern. The large aggregate of mitochondria was prominent in in vitro matured oocytes than in vivo MII oocytes.
The intensities of florescent (Figure 4) in GV oocyte in non-vitrified (fresh) and vitrified groups were 48.47±1.8 and 38.69±1.1 and in IVM-MII and OV-MII oocytes in non-vitrified group were 47.21±1.4 and 44.43± and in IVM-MII and OV-MII of vitrified group were 41.60±1 and 34.05±3.9, respectively. These rates were significantly different between vitrified and non-vitrified groups and between in vitro matured group and in vivo groups (p<0.05).
Discussion :
This study confirmed that cryotop method is suitable for vitrification of mouse oocytes at GV and MII stages 17,18. Vitrification of oocytes at different developmental stages did not affect the subsequent development of oocytes, however, there were some controversial results in this regard. Diversity of the examined species, cryoprotectant solution and carrier system could result in some differences in findings. Wu et al using the cryotop method showed that survival, nuclear maturation and subsequent development of porcine oocytes may be affected by vitrification according to their stage of maturation at the time of vitrification 19. Several studies indicated that the quality of vitrified oocytes varied using similar carrier system depending on the type of cryoprotectants 20,21.
Moreover, in the present study, an attempt was made to evaluate the effects of three factors on the ATP content of oocyte including developmental stage of oocytes, in vitro culture and vitrification and warming procedure.
Our observations indicated that ATP content of mouse oocytes at GV and MII did not significantly change. The level of ATP related to its production and consumption, the number and ultrastructure of mitochondria. The balance between these three basic factors in each developmental stage may cause ATP content to be constant. Similarly Van Blerkom et al showed that the average ATP content of GV and MII stages of mouse oocytes was constant 22. In contrast, it was shown that ATP level of bovine 10 and pig 23 MII oocytes contain more ATP than their immature oocytes. This may be due to the diversity of the examined species, and/or the IVM conditions which were used.
In other part of the present study, it was shown that the level of ATP in MII oocytes was similar in both in vitro and in vivo groups. This finding may be related to appropriate culture condition. Two main factors may affect the ATP content of oocytes; (a) intrinsic metabolic factors that are oocyte-specific and (b) extrinsic factors. These factors influence the oocyte maturation in in vivo and in vitro condition 24. Chi et al suggested that reduced metabolism may be due to intrinsic factors rather than a response to suboptimal culture conditions 25. There are other studies which demonstrated that concentration of ATP was not influenced by different maturation conditions 22,23,26.
Our observation showed that vitrification method used in this study did not affect ATP content of oocytes in both GV and MII oocytes and it is the first comparative study on ATP content of oocytes at GV and MII stages after vitrification and warming procedures. It was proposed that incubation after warming allowed oocytes to regain some ATP level 27-29. Consistent with our results, Chankitisakul et al reported that vitrification by cryotop did not reduce ATP content in bovine oocytes at MII stages 30. However, some researchers reported that vitrification/warming has a significant negative effect on oocyte ATP concentration 27-29.
Differences in reported oocyte ATP content in different studies may be due to differences in studied species, their genetics, age, superovulation protocols, culture medium, vitrification protocol and the method of ATP content measurement.
The differences between the florescent intensity of oocytes were observed not only in vitrified and non-vitrified groups but also between in vitro matured oocyte and in vivo groups and it could be related to different number and pattern of mitochondria of oocytes. It was proposed that this difference in appearance of mitochondria between in vitro and in vivo condition may be caused by some alteration in culture condition including temperature (thermal shock), pH alteration, ROS production, osmolarity changes, which require more investigations.
In previous reports, different patterns of mitochondrial distribution in several mammals' oocytes were reported 5,10,11,31. Diversity of studied species and varying degrees of cytoplasmic maturation of oocytes may be responsible for these controversies.
Conclusion :
In summary, our findings showed that cryotop vitrification method using EG and DMSO and sucrose as cryoprotectant didn’t affect the developmental competence and ATP content of mouse oocyte but some alteration was seen in mitochondria distribution of in vitro matured oocytes in comparison to their controls.
Acknowledgement :
We warmly thank Mr. Sharam Pourbeyranvand and Mrs. Saideh Ebrahimi for photographic assistance. All authors contributed to the study design and to drafting the paper. Prof. Mojdeh Salehnia directed the study together with Zohreh Nazmara. Dr. Saman HosseinKhani provided some laboratory facilities and excellent scientific comments for ATP assay. This study was supported by Tarbiat Modares University.
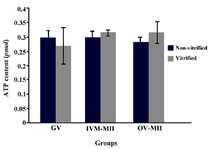
Figure 1. The adenosine triphosphate (ATP) content of mouse oocyte at different developmental stages in vitrified and non-vitrified groups and in vitro matured oocytes. There was not any difference between the groups. IVM-MII: MII oocyte derived from in vitro maturation of GV oocyte and OV-MII: MII oocyte collected from mouse oviduct
|
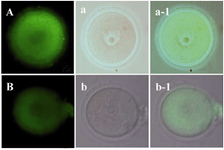
Figure 2. Distribution of mitochondria stained by Mito Tracker Green in A) non-vitrified and B) vitrified GV oocytes. The phase contrast micrograph of the same groups was shown in the second column (a and b). The merge of figures was shown in the third column
|
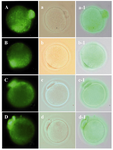
Figure 3. The pattern of oocyte mitochondria stained by Mito Tracker Green was shown in; A) MII oocyte derived from in vitro maturation of non-vitrified GV oocyte, B) MII oocyte derived from in vitro maturation of vitrified GV oocyte, C) Non-vitrified MII oocytes and D) Vitrified MII oocytes after warming. The phase contrast micrograph of the same groups was shown in the second column, respectively (a-d). The merge of figures was shown in the third column
|
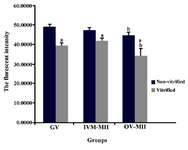
Figure 4. The fluorescence intensity of oocytes stained by Mito Tracker Green at GV and MII stages in non-vitrified and vitrified groups. GV: germinal vesicle oocytes; IVM-MII: MII oocyte derived from in vitro maturation of GV oocyte and OV-MII: MII oocyte collected from mouse oviduct. a) significant difference with non-vitrified group; b) significant difference with IVM-MII groups (p<0.05)
|
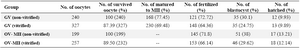
Table 1. The survival and developmental rates of vitrified and non-vitrified oocytes at different developmental stages
|
|