Ectopic Expression of Sortilin 1 (NTR-3) in Patients with Ovarian Carcinoma
-
Hemmati, Shayda
-
Monoclonal Antibody Research Center, Avicenna Research Institute, ACECR , Tehran, Iran
-
Department of Cell and Molecular Biology, Khatam University , Tehran, Iran
-
Zarnani, Amir-Hassan
-
Nanobiotechnology Research Center, Avicenna Research Institute , Tehran, Iran
-
Immunology Research Center, Faculty of Medicine, Iran University of Medical Sciences , Tehran, Iran
-
Mahmoudi, Ahmad Reza
-
Monoclonal Antibody Research Center, Avicenna Research Institute, ACECR , Tehran, Iran
-
Sadeghi, Mohammad Reza
-
Reproductive Biotechnology Research Center, Avicenna Research Institute, ACECR , Tehran, Iran
-
Soltanghoraee, Haleh
-
Reproductive Biotechnology Research Center, Avicenna Research Institute, ACECR , Tehran, Iran
-
Akhondi, Mohammad Mehdi
-
Reproductive Biotechnology Research Center, Avicenna Research Institute, ACECR , Tehran, Iran
-
Tarahomi, Majid
-
Reproductive Biotechnology Research Center, Avicenna Research Institute, ACECR, Tehran, Iran
-
Jeddi-Tehrani, Mahmood
-
Monoclonal Antibody Research Center, Avicenna Research Institute, ACECR , Tehran, Iran
-
Immune and Gene Therapy Lab, CCK, Department of Oncology-Pathology, Karolinska University Hospital Solna, Karolinska Institutet , Stockholm, Sweden
-
 Rabbani, Hodjattallah
Ph.D., Department of Antigen and Antibody Engineering, Monoclonal Antibody Research Center, Avicenna Research Institute, ACECR, Tehran, Iran, P.O. Box: 19615-1177, Tel: +98-21-22432020, Fax: +98-21-22432021, E-mail: hodrab@ki.se
Rabbani, Hodjattallah
Ph.D., Department of Antigen and Antibody Engineering, Monoclonal Antibody Research Center, Avicenna Research Institute, ACECR, Tehran, Iran, P.O. Box: 19615-1177, Tel: +98-21-22432020, Fax: +98-21-22432021, E-mail: hodrab@ki.se
-
Monoclonal Antibody Research Center, Avicenna Research Institute, ACECR , Tehran, Iran
-
Immune and Gene Therapy Lab, CCK, Department of Oncology-Pathology, Karolinska University Hospital Solna, Karolinska Institutet , Stockholm, Sweden
Abstract: Gene expression profiling of ovarian carcinoma tissues has shown an increase of four-fold expression of SORT1 gene. Sortilin 1 (NTR-3) is a 95-100 kDa protein normally expressed in heart, brain, placenta, skeletal muscle, spinal cord, thyroid, and testis. However, its expression has never been reported in normal ovary. Here, we report expression of sortilin 1 in ovarian carcinoma tissues both at gene and protein levels. Sortilin 1 was expressed in all ovarian carcinoma patients (n=15) as well as ovarian carcinoma cell lines (n=5) regardless of their phenotypic characteristics. Non-malignant ovaries (n=6) did not express sortilin 1. The molecular basis for this ectopic expression is not yet clear. Our results showed a major cell surface expression of sortilin 1 rather than ER-Golgi compartment where it is mainly expressed. This finding may introduce sortilin 1 as a novel tumor marker for diagnosis of ovarian carcinoma and may signify its therapeutic value in targeted therapy.
Introduction :
Ovarian cancer is one of the leading causes of cancer deaths in women. Due to the lack of any specific tumor marker, the diagnosis of this disease usually happens in advanced stages leading to high mortality rate. Al-though, the increased level of CA125 is con-sidered to be a diagnostic marker (in about 80% of cases), but it is still not reliable and usually increases at very late stages. Since no suitable diagnostic and curative therapy is available, there is a high demand for finding novel tumor markers in this malignancy both for diagnostic and therapeutic purposes.
Gene expression profiling of ovarian car-cinoma patients in early and late stages or with low malignant potential has shown a nearly four-fold increase of SORT1 gene ex-pression (1).
The human SORT1 gene consists of 22 exons located on the short arm of chrom-osome 1 (1p21.3-p13.1) encoding a type I receptor of 831 amino acids with a molecular weight of 95-100 kDa. Seven non-coding transcript variants for this gene have also been reported.
SORT1 is expressed at gene level in heart, brain, placenta, skeletal muscle, spinal cord, thyroid, and testis (2). Its expression, however, has never been reported in normal ovary. Sortilin 1 is a member of family of Vps10p-domain receptors. It is known as a multi-ligand molecule and such proteins as lipo-protein lipase (LPL), Neurotensin 3 (NT3), Receptor associated protein (RAP), proNGF, and thyroglobulin are among its potential ligands (2, 3-5).
The function of sortilin 1 varies depending on its location and the presence of its ligands. Sortilin 1 acts as a sorting receptor when it is expressed in trans-golgi network and also as a clearance receptor when it is expressed on cell surface (2, 3-5).
The role of sortilin 1 and NT3 as its ligand in cancer cell growth has previously been reported (6). Although, sortilin 1 as a type I receptor lacks the necessary domains involved in signaling, but is an essential part of a re-ceptor complex for pro-NGF induced neural cell death (5, 7).
The four-fold overexpression of SORT1 in ovarian carcinoma together with its driving role in cancer cell growth brings forward the potential involvement of this protein in the pathobiology of this disorder. To test this hy-pothesis, we studied the expression of sortilin 1 in 15 ovarian carcinoma patients both at gene and protein levels. Sortilin 1 was expres-sed in all 15 ovarian carcinoma tumor tissues as well as related cell lines, while no expres-sion of this molecule in non-malignant ovarian tissue was found. The expression of sortilin 1 in ovarian carcinoma patients may represent a crucial role of this protein in tumorigenesis driving machinery.
Materials and Methods :
Patients and tissue samples
Ovarian tissue samples were obtained from patients at Imam Khomeini Hospital. After a consent letter was signed by each patient, all the aspects of this study were approved by the Avicenna Local Ethics Committee. After surgical resection, each fresh tumor specimen was immediately divided into two portions: one portion was instantly frozen in liquid nitrogen and the other was OCT (Optimal Cutting Compound) embedded using Jung freezing medium (Leica Instruments, Nussloch, Germany) for immunohistochem-ical investigations.
Fifteen malignant ovarian tissue samples including papillary serous carcinomas (n=9; mean age 48.3±16.08 yr), endometrioid carcinoma (n=1; age 39), mucinous carcin-omas (n=2; age 59 and 70 yrs), clear cell carcinoma (n=1; age 45 yr), germ cell tumor (n=1; age 28 yr), and sex cord-stroma tumor (n=1; age 22 yr) were investigated. A total of six non-malignant ovarian tissues (mean age 45.3±4.2 yr) served as control group (Table 1). Tissue sections made from each sample, stained with Hematoxylin and Eosin (H&E) and examined by two independent pathologists to confirm the pathological state of each sample.
Cell lines
The human ovarian carcinoma cell lines (Table 2) were cultured in RPMI 1640 (Gibco, Invitrogen, USA), containing 10% fetal bovine serum (Biochrom AG, Berlin, Germany), 100 units/ml penicillin (ICN Bio-medicals, Ohio, USA), and 100 µg/ml strepto-mycin (Sigma, St Louis, MO). All cell lines were cultured at 37 °C in a humidified incu-bator with 5% CO2 atmosphere.
RNA extraction and cDNA synthesis
Total RNA was extracted from cell lines and tissue samples using RNA-Bee reagent (BioSite, Täby, Sweden) according to the manufacturer’s instruction. The quality of the RNA samples was determined by agarose gel electrophoresis after staining with ethidium bromide, and visualization under UV light. Total RNA was unfolded at 65 °C for 10 min. cDNA was then synthesized using 5 µg of total RNA in 20 µL reaction mixture consisting of 4 µL of 5x reaction buffer, 1 µL of 10 mM dNTPs, 1 µL of 10 pmol/mL random hexamers (N6), and 200 U M-MuLV reverse transcriptase (Fermentas GmbH, St. Leon-Rot, Germany). The reaction mixture was incubated at 42 °C for 45 min.
RT-PCR amplification of SORT1
RT-PCR amplification was performed using SORT1 gene (g.b. NM_002959) specific primers of “CAGTCCAAGCTATATCGAA GTGAGG” as sense and “AAGATGGTGTT GTCTGATCCCCATTT” as antisense (Ope-ron, Köln, Germany). Briefly, 25 µL reaction mixture of PCR was prepared using 2.5 µL 10x buffer, 3 µL 25 mM MgCl2, 1 µL dNTPs (10 mM), 5 pmol of each primer, 1 Unit Taq DNA polymerase (Roche, Germany) and 1 ?l template cDNA. PCR was then followed by 35 cycles (for SORT1) or 30 cycles (for ß-actin) at 92 °C for 30 sec, 60 °C for 30 sec, and 72 °C for 30 sec leading to a 383 bp SORT1 or 155-bp ß-actin amplicons. To ensure the specificity of primers, some of the SORT1 PCR products were cloned into pGEM-T easy vector (Promega, Madison, WI) and were subjected to sequencing.
Due to the poor quality of RNA prepared from some tissue samples, nested-PCR was carried out for detection of SORT1 gene in ovary tissues using “GTTAACAGCAGAG GTGTCTGGAG” as sense primer and “AA ACATACTGCTTTGTGGATTTC” as anti-sense primer. Briefly, 1 ?l of RT-PCR prod-uct was added to a new 24 ?l reaction mixture containing 2.5 ?l 10x PCR buffer, 3 ?l 25 mM MgCl2, 1 ?l 10 mM dNTPs, 5 pmol of each primer, and 1 Unit Taq DNA polymerase. Nested PCR was performed for 20 cycles generating an amplicon of 225 bp. PCR products were finally visualized by running agarose gel electrophoresis containing ethi-dium bromide.
Western blotting
Tissues and cell lines were lysed in lysis buffer containing 2% Triton X-100, 10 mM Tris, pH 7.4, 100 mM NaCl, 1 mM EDTA, 1 mM NaF, 20 mM Na4P2O7, 1% glycerol, 0.1% sodium dodecyl sulfate (SDS), and 1% protease inhibitor cocktail (Sigma) on crashe
Result :
SORT1 gene expression
Representative RT-PCR gel of ovarian carcinoma patients is shown in Figure 1. All ovarian carcinoma patients (n=15) expressed SORT1 at the mRNA level (Table 1). SORT1 was also expressed in all six ovarian carcin-oma cell lines (Table 2). Non-malignant ovary, however, failed to express this gene (data not shown).
Sortilin 1 protein expression
Western blot analyses of six ovarian carcinoma cell lines showed a band of 95-100 kDa as well as an extra band of 50 kDa (Figure 2A). Cell lysates from primary ovarian carcinoma tissues of different subtypes showed only a band of 95-100 kDa of sortilin 1 (Figure 2B). None of the non-malignant ovary samples expressed sortilin 1 at protein level (Figure 2C).
To confirm the expression and localization of sortilin 1 in tumor samples, we performed indirect immunofluorescence staining on 7 malignant ovarian tissue samples and 5 normal ovaries. The positive staining was only observed in the cell membrane of malignant epithelial cells. Consistent with the results obtained by Western blotting, the ovarian carcinomas (four of seven specimens) expressed sortilin 1 mainly localized to cell surface rather than ER-Golgi compartment in immunohistochemistry & tainins. Three out of seven ovarian carcinoma samples showed lower levels of sortilin 1 in immunofluores-cence experiments. Similar to Western blot results, no sortilin 1 expression was observed in normal ovary tissue sections. One represen-tative out of seven immunofluorescence ex-periments is shown in Figure 3.
Discussion :
The functional roles of neuropeptides (NT) and their receptors in cancer cell growth are now well established. The growth effect of sortilin 1 as neurotensin-3 receptor (NTR-3) in prostate, colon, and pancreatic cancer cell lines strongly suggests the involvement of this protein in cell growth (8). Sortilin 1 was also expressed in a panel of breast cancer cell lines (our unpublished data). It seems that the ex-pression of sortilin 1 is not restricted to ovarian carcinoma and it might be also ex-pressed in other solid tumors. This may sig-nify more important function of this receptor in cancer development.
In our study, the cell lysates from all ovarian cancer tissues and cell lines but not from non-malignant ovary tissues, showed expression of 95-100 kDa sortilin 1 protein indicating an ectopic expression. Interesting-ly, a 50 kDa band was only observed in ovari-an carcinoma cell lines whereas it was absent in breast cancer cell lines (data not shown). This 50 kDa band may represent an uniden-tified isoform of sortilin 1 which requires further characterization.
Although, sortilin 1 is mainly localized to ER-Golgi apparatus, it is also expressed as a mature cell surface receptor (9). Our immuno-fluorescence experiments showed that sortilin 1 is mainly expressed on tumor cell surface rather than ER-Golgi compartment. The sur-face expression of this protein may provide a more significant role for this protein in ERK signaling pathway (10) leading to proliferation of cancer cells. Apparently, expression of sortilin 1 in ovarian cancer cells as a cell sur-face receptor does not induce apoptosis ex-cluding its proapoptotic activity. This might be due to the lack of other co-receptors re-quired for triggering apoptosis in ovary cancer cells and the absence of their signaling mediators (proNGF, proBDNF). It could be also due to structural differences in sortilin 1 expressed by cancer cells leading to impaired heterodimerization with other co-receptors (p75NTR) which is necessary for signal trans-duction. In general, the dual functional pro-apoptotic and anti-apoptotic roles of sortilin 1 must be considered in normal and pathogenic conditions.
Among patients enrolled in this study two patients had tumors with morphology of non-epithelial origin indicating that expression of sortilin 1 is not restricted to epithelial cells. Due to the lack of patient samples from early stages and also limited number of patients, it was not possible to correlate the expression level of sortilin 1 to the stage of the disease as well as to CA125 level or age of the patients. In this regard the expansion of the study in more patients is warranted.
Recent report shows that sortilin 1 is also expressed in thyroid epithelial cells capable of binding with high affinity to thyroglobulin, another evidence for sortilin 1 as a multi-ligand receptor (4). This finding together with the expression of sortilin 1 in ovarian carcin-oma may explain the 4% incidence of metas-tasis of ovarian carcinoma to thyroid glands, rich in thyroglobulin (11) as well as the same incidence in brain metastases (12).
Altogether, ectopic expression of sortilin 1in ovarian carcinoma tissues, its absence in normal human ovary, its involvement in ERK signaling pathway (10) as well as its restricted tissue expression in women makes it a poten-tial candidate for developing more selective targeted therapy approach in patients with ovarian carcinoma. In this regard, generation of monoclonal antibodies capable of inducing apoptosis and also competing with sortilin 1 ligand binding site (Vps10p domain) is recommended.
Women with cancer may suffer from infertility after medical treatment. The current approach to protect reproductive capability in women with cancer is ovarian cortex cryo-preservation prior to any medical treatment and re-implanting the frozen ovary tissues after cancer therapy. Detection of sortilin 1, as a unique marker in the cryopreserved tissue<
Acknowledgement :
This work was supported by grants from Presidential Science and Technology Deputy of Islamic Republic of Iran. We thank Mrs. Zohre Sadeghian (Imam Khomeini Hospital, Tehran, Iran) for her assistance in collecting tissue samples.
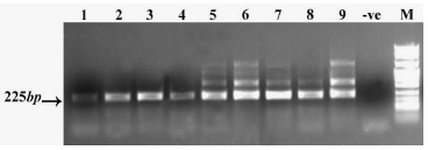
Figure 1. SORT1-specific mRNA expression (nested RT-PCR) in ovarian cancer tissue samples. Lanes 1-9 tissue samples, -ve: Negative control PCR reaction without template, M: DNA size marker. The figure represents 9 experiments out of 15. The extra bands over 225 bp are due to the overload of primary PCR reactions
|
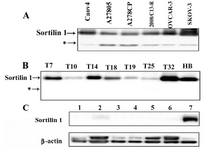
Figure 2. Western blot analysis of sortilin 1 using rabbit polyclonal anti-sortilin antibody under non-reducing conditions. A) All ovarian cancer cell lines exhibited expression of about 100 kDa sortilin 1 protein. *A band of about 50 kDa was also expressed by these cell lines. B) Ovarian cancer tissue samples T7, T10, T14, T18, T19, T25, and T32 corresponding to patients in Table I selected based on their differentiation. Human brain (HB) cell lysate served as positive control. C) Lanes 1-6 representing non-malignant ovarian tissue samples. Lane 7 represents human brain cell lysate. β-actin expression was used as an internal loading control
|
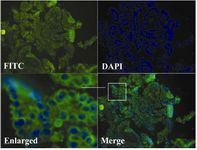
Figure 3. Immunofluorecense staining of sortilin 1 in ovarian carcinoma tissue (T15) using rabbit anti-sortilin 1 polyclonal antibody. The enlarged part clearly shows the cell surface expression of sortilin 1. The figure represents one experiment out of seven
|
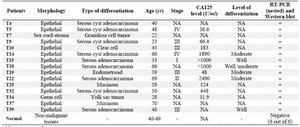
Table 1. Expression of sortilin 1 and phenotypic characterization of 15 ovarian carcinoma patients compared to the normal ovary tissues from 6 healthy individuals
NA: not assigned
|
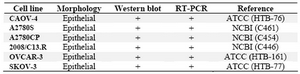
Table 2. Expression of sortilin 1 in ovarian cancer cell lines
NCBI; National Cell Bank of Iran, ATCC; American Type Culture Collection
|
|