Development of a High Sensitive Multiplex Lateral Flow Immunoassay (LFIA) System for Rapid Detection of Methicillin-Resistant Staphylococcus Aureus (MRSA)
-
Amini, Masoomeh
-
Department of Pathobiology, School of Public Health, Tehran University of Medical Sciences, Tehran, Iran
-
 Pourmand , Mohammad Reza
Department of Pathobiology, School of Public Health, Tehran University of Medical Sciences, Tehran, Iran, Tel: +98 9123933624; Fax: +98 21 88991117; E-mail: mpourmand@tums.ac.ir
Pourmand , Mohammad Reza
Department of Pathobiology, School of Public Health, Tehran University of Medical Sciences, Tehran, Iran, Tel: +98 9123933624; Fax: +98 21 88991117; E-mail: mpourmand@tums.ac.ir
-
 Faridi-Majidi , Reza
Department of Medical Nanotechnology, School of Advanced Technologies in Medicine, University of Medical Sciences (TUMS), Tehran, Iran, E-mail: refaridi@sina.tums.ac.ir
Faridi-Majidi , Reza
Department of Medical Nanotechnology, School of Advanced Technologies in Medicine, University of Medical Sciences (TUMS), Tehran, Iran, E-mail: refaridi@sina.tums.ac.ir
Abstract: Background: Methicillin-resistant Staphylococcus aureus (MRSA) has become a worldwide concern as an epidemic bacterium and a cause of nosocomial and community-acquired infections. One of the major problems in the prevention and treatment of infections caused by MRSA strains is their multi-drug resistant trait, which causes the spread of infections and increases the mortality rate. Therefore, a rapid and accurate method is needed to identify MRSA strains, initiate appropriate antibiotic therapy, and control its infection. The aim of this study was to develop a twin lateral flow immunoassay system to detect methicillin-resistant Staphylococcus aureus (MRSA).
Methods: First, BSA blocked AuNPs-anti-peptidoglycan antibody and AuNPs-anti-BSA antibody were used to detect Staphylococcus aureus (S. aureus). Then, AuNPs-anti-PBP2a antibody was used to specifically detect MRSA. Sensitivity, specificity and limit of detection of this twin immunoassay system were assessed using MRSA, methicillin susceptible S. aureus and clinical samples. Results were compared to those of cefoxitin disc diffusion (FOX30) and Polymerase Chain Reaction (PCR) as gold standards.
Results: The Limit of Detection (LOD) of this twin system were 103 and 104 CFU/ml for the first and second strips, respectively. Sensitivity and specificity of this innovative assay in detecting MRSA were 92.30 and 97.36%, compared to FOX30 and PCR, respectively.
Conclusion: High rates of sensitivity and specificity of this initiative system show its high potentials for rapid and accurate detection of MRSA.
Introduction :
Methicillin-resistant Staphylococcus aureus (MRSA) has become a worldwide concern as an epidemic bacterium and a cause of nosocomial and community-acquired infections 1. This species causes a wide variety of diseases, foodborne infections, mild skin infections, and potentially fatal diseases including pneumonia and sever sepsis 2. Technically, treatment of MRSA is a challenge. Vancomycin belongs to the glycopeptide group of antibiotics and has been used for the treatment of MRSA infections for more than 50 years. However, there are significant concerns in use of this antibiotic in infections by Staphylococcus aureus (S. aureus). Linezolid, Daptomycin and ceftaroline have been approved for the treatment of infections by drug-resistant Gram-positive pathogens. They also have significant limitations. Resistance to methicillin in MRSA strains is related to the production of a modified penicillin-binding protein 2 (PBP2 or PBP2a) and an enzyme involved in the final step of the peptidoglycan synthesis with a molecular mass of 76 kDa consisting of 668 amino acids encoded by the mecA gene 3. One of the major problems in the prevention and treatment of infections caused by MRSA strains is their multi-drug resistant trait, which causes the spread of infections and increases the mortality rate. Therefore, a rapid and accurate method is needed to identify MRSA strains, initiate appropriate antibiotic therapy, and control its infection 3,4.
Conventional methods for the detection of MRSA isolates are based on phenotypic and genotypic characteristics of the bacterial isolates. Despite their advantages, these methods have several limitations including time, cost, and material consumption as well as the need for many prerequisites, trained personnel, and sophisticated equipment 5, which are not practical for routine tests and daily use in clinical laboratories 6,7. Therefore, it is crucial to develop a cheap, fast, and powerful diagnostic method to detect Point of Care (POC) MRSA. Nanotechnology has been potentially used in diagnosis of diseases. Developments in nanotechnology have led to novel nanomaterials in biomedical applications. Common biomedical materials include liposomes, polymeric micelles, graphene, carbon nanotubes, quantum dots and gold nanoparticles (AuNPs). Of these materials, AuNPs have been addressed as the most interesting nanomaterials because of their unique optical, sensing and biochemical characteristics. Recently, AuNPs have been used in drug deliveries and microbial infection therapies.
Lateral Flow Immunoassay (LFIA), a single-step diagnostic method, has become the most practical platform for POC detection by delivering results in a short time, requiring a small sample volume(s), eliminating the need for trained staff, which is very cost-effective 8. The few studies conducted on the capability of LFIA strip test to detect MRSA have mostly relied on colloidal gold-labeled monoclonal antibody technology. They revealed that their antigenic targets are not suitable for this technique or their detection limit is low 9-13, indicating that the signal amplification is the bottle neck of the construction of a high sensitive LFIA detection system. Hence, more delicate designs with higher sensitivity and specificity are needed to diagnose such infectious agents.
In the present study, the LFIA signal was intensified by a multiplex system to detect MRSA. This approach has some advanced features compared to approaches used in previous studies. The diagnostic system had a multi-strip design. The first strip was pre-screening the S. aureus isolates, where a monoclonal antibody was used against cell-wall peptidoglycan of S. aureus. In this strip, a dual labeled detector element was used to amplify the detection signal. The second strip was constructed to detect MRSA among the pre-screened S. aureus. In this strip, the anti-PBP2a (6G10) monoclonal antibody (McAb-AuNP), considered as MRSA specific biomarker, was used. The use of a dual conjugated pad along with two specific antigenic targets led to the development of a novel, highly-sensitive, and specific LFIA system for MRSA detection. The present study was designed for the first time to detect MRSA isolates. The results could show a great potential for converting as a POC testing for early detection of infection in medical centers and laboratories.
Materials and Methods :
Antibodies, reagents and requirements: Bacterial strains and clinical isolates used in this study (Table 1) were obtained from two hospitals of Tehran University of Medical Sciences. Two different clones of PBP2a monoclonal antibody, (clone 6G10 and clone 17C8) were purchased form Abnova, Taiwan, and used as a conjugated and capturing antibody, respectively. Goat anti-mouse IgG polyclonal antibody was obtained from BBI Solutions, UK. Anti-peptidoglycan S. aureus was purchased from Abcam, UK. Bovine Serum Albumin (BSA), monoclonal Anti-BSA antibody, HAuCl4, sucrose, sodium citrate and casein was obtained from Sigma-Aldrich, USA. All components of lateral flow test, including a Polyvinyl Alcohol (PVA), backing pad, conjugate pad, absorbent pad and sample pad were purchased from Jiening Biotech, China. Nitrocellulose (NC) membrane was prepared from Millipore, USA. Nano Drop 2000 UV-Vis Spectrophotometer (Termo Scientific, USA) was used to obtain spectra from AuNPs and conjugates. Transmission electron microscopy (TEM; 2100 F 200 kV TEM, JEOL, USA) and Dynamic Light Scattering (DLS) was used to analyze the size and shape of synthesized AuNPs.
Bacterial strains
Clinical samples and bacterial identification: Totally, 166 S. aureus clinical isolates were randomly identified from 500 different clinical specimens collected from hospitals of Tehran University of Medical Sciences, Tehran, Iran. First, patients signed written informed consents. Then, clinical specimens, including wound swabs, urine, Bronchoalveolar Lavage (BAL), abscess, exudates from lesions and tissue biopsies, were immediately transferred to Tehran University of Medical Sciences Molecular Laboratory. Samples were cultured on blood agar and MacConkey agar and incubated for 24 hr at 37°C and then identified using phenotypic and genotypic assays 5. The methicillin resistance of the isolates were identified by cefoxitin disc diffusion (30 μg, MAST Diagnostics, UK) on Mueller-Hinton agar (MHA), according to the Clinical and Laboratory Standards Institute (CLSI) guideline 5.
Molecular identification of MRSA: A) DNA extraction: Genomic DNA was extracted from MRSA isolates using genomic DNA extraction kit (YTA, Iran) according to the manufacturer’s instruction.
B) PCR amplification of mecA gene: The presence of mecA gene in samples was confirmed by PCR using pair of primers of mecAF1 (5’-TGCTATCCACCCTC AAACAGG-3’) and mecAR1 (5’-AACGTTGTAACC ACCCCAAGA-3’). Briefly, PCR reaction was performed using Eppendorf Termocycler (Eppendorf, Germany) in 25-μl reactions, including 2 μl of DNA templates, 0.25 μl of each primer (10 µM), 12.5 μl master mix (SinaClon, Iran), 12.5 μl MgCl2 and 8.3 μl sterile DDW. The PCR program was as follows: initial denaturation at 94°C for 2 min, 35 cycles of denaturation (94°C for 2 min), annealing (60°C for 1:30 min), extension (72°C for 2 min) and a final extension at 72°C for 2 min. Methicillin resistant (COL) and standard S. aureus ATCC 8325-4 were used as positive and negative controls, respectively.
Synthesis of colloidal gold nanoparticles (AuNPs): AuNPs in average optimized size of 14±2 nm 14 were synthesized according to reversed Turkevich method 15{Faridi Majidi, 2019 #34;Dong, 2020 #20}. In brief, 25 ml of DDW were added into Erlenmeyer flask and heated to boiling point. At 90°C, 11.5 µl of 0.01% (m/v) Chloroauric acid (HAuCl4) with concentration of 250 mg/ml was added with vigorous stirring and the solution was heated to reach boiling point. After 10 min, 650 µl of sodium citrate solution (4% w/v) was added to the center of the flask at once under stirring. Boiling and stirring was continued for 15 min; during the procedure, the solution color changed from light yellow to wine red. Then the solution was left to cool and stirring was continued for an additional 15 min. Size, morphology and polydispersity of the AuNPs were determined by Transmission Electron Microscopy (TEM) and Dynamic Light Scattering (DLS), respectively. The lambda max (λ max) of the synthesized AuNPs was measured at wavelengths ranging from 400 to 600 nm by ultraviolet-visible (UV/vis) spectrophotometry.
Preparation of the AuNPs conjugates
AuNPs conjugates for peptidoglycan strip (the first strip): A) AuNPs conjugates at the first layer: To prepare the first AuNPs conjugate, the pH value of the colloidal gold solution (1%, w/v) was adjusted to 9.5 with NaOH (0.1 M). Anti-peptidoglycan S. aureus McAb (10 μl, 1.6 μg/ml) was added to 450 ml pH-adjusted colloid gold solution. The mixture was gently stirred for 30 min and the McAb-AuNPs conjugate was further stabilized. Then, 50 μl of BSA solution (with a final concentration of 0.1% w/v) was added to the solution and incubated at room temperature with gentle stirring for 30 min and the mixture was centrifuged at 10000 rpm for 50 min at 4°C. Then, the colloidal gold pellets were resuspended in 500 μl borate buffer (2 mM, pH=7.5), containing 4% sucrose, 6% trehalose and 0.05% (w/v) sodium azide. The final McAb-AuNPs conjugates were stored at 4°C. OD of the conjugates was assessed as reported previously 14.
B) AuNPs conjugates at the second layer: To prepare the second AuNPs conjugates, the pH value of the colloidal gold solution (1% w/v) was adjusted to 9.5 with NaOH (0.1 M). Anti-BSA McAb (2 μl, 4 μg/ml) was added to 450 ml pH-adjusted colloid gold solution. The mixture was gently stirred for 30 min and to block the nonspecific binding between the McAb-AuNPs, thereafter, 50 μl Casein solution (with a final concentration of 0.1%) was added to the solution and incubated at room temperature with gentle stirring for 30 min. The mixture was centrifuged at 10000 rpm for 40 min at 4°C. Then the colloidal gold pellets were re-suspended in 500 μl borate buffer (2 mM, pH=7.5) containing 4% sucrose, 6% trehalose and 0.05% (w/v) sodium azide. The final McAb-AuNPs conjugates were stored at 4°C. OD of the conjugates was assessed as reported previously 14. To compare, an LFIA strip without the second conjugate pad in the first strip was also prepared, and the sample pad was attached to the first conjugate pad.
AuNPs conjugates for PBP2a strip (the second strip): The NaOH solution (0.1 M) was used for adjusting the pH of the colloidal gold solution (1% w/v) to 9.5 and 3.3 μl anti-PBP2a McAb (6G10), (40 μg/ml) was added to 450 ml pH-adjusted colloidal gold solution. The mixture was gently stirred for 30 min and to block the nonspecific binding between the McAb-AuNPs; then, 50 μl BSA solution (with a final concentration of 0.1% w/v) was added to the solution, incubated at room temperature and gently stirred for 30 min. The mixture was centrifuged for 45 min (at 4°C and 10000 rpm). After centrifugation, the supernatant was discarded, and 500 μl borate buffer (2 mM, pH=7.5) containing 4% sucrose, 6% trehalose and 0.05% (w/v) sodium azide was added to AuNPs conjugate to be re-suspended. The final McAb-AuNPs conjugates were stored at 4°C.
Assembling of the LFIA strips
Peptidoglycan strip (S. aureus specific strip): Peptidoglycan strip (S. aureus specific strip) is comprised of a sample pad, two conjugate pads, Nitrocellulose (NC) membrane, and an absorbent pad as shown in figure 1A. NC membrane was first pasted onto an adhesive backing card. Then, 0.5 μl of anti-peptidoglycan McAb (1.1 μg/μl in PBS buffer, pH=7.4) and 0.5 μl goat anti-mouse IgG (1 μg/μl in PBS buffer, pH=7.4) were dropped to a strip of NC membrane as a test and control dots, respectively. Subsequently, the NC membrane was dried in a desiccator at room temperature. The sample and conjugate pad were soaked in aqueous solution (50 mM borate buffer, pH=7.5 containing 5% trehalose), Then dried in incubator and stored in desiccators at room temperature. Two conjugate pads were soaked with a desired volume of first and second AuNPs conjugates. First, AuNPs conjugate including AuNPs-anti-peptidoglycan McAb was blocked with BSA, and the second, including anti-BSA McAb was blocked with casein and placed in different pads on a one strip. Afterwards the sample pad was attached to the second AuNPs conjugate. The pads were then dried at room temperature and stored in a desiccator at 4°C. All of the components were mounted on a backing card with a 2 mm overlap. To check the performance, we have prepared a control LFIA strip only with first AuNPs conjugate.
PBP2a strip (MRSA specific strip): To construct PBP2a strip (MRSA specific strip) (Figure 1B), the NC membrane was first pasted onto an adhesive backing card. The anti-PBP2a McAb (6G10)– AuNPs conjugate pad was pasted on the adhesive backing card with a 2 mm overlap with the end of the NC membrane. The sample pad with a 2 mm overlap with conjugate pad and absorbent pad with a 2 mm overlap with NC membrane were mounted on a backing card. Anti-PBP2a McAb (17C8) (1.1 μg/μl in PBS buffer, pH=7.4) and goat anti-mouse IgG (1 μg/μl in PBS buffer, pH=7.4) at a volume of 0.5 μl per dot were used in NC membrane as the test and control dots, respectively.
Limit of Detection (LOD) calculation: For in-vitro evaluation of LOD, 100 μl serially diluted MRSA strain (COL) and methicillin susceptible S. aureus ATCC 8325-4 suspended in PBS (100–107 CFU/ml) were applied to the sample pad and visually evaluated. The PBS buffer was applied to the sample pad as negative control. After 15 min, test signals were developed. The LOD of the prepared Staphylococcus Flow Immuno Assay (SFIA) test strips was calculated. Test line signals were measured by Image J 1.52i Software.
In-vitro evaluation of sensitivity and specificity of LFIA test strips: For in vitro evaluation of sensitivity and specificity of LFIA, 100 μl of clinical samples serially diluted in 1× PBS buffer (100–107 CFU/ml) and 100 μl of strains other than Staphylococcus listed in table 1 at a concentration of 107 CFU/ml, were individually applied to the sample pad and visually evaluated. The PBS buffer and MRSA (COL strain) suspension (107 CFU/ml in PBS) were applied to the sample pad as negative and positive control, respectively. After 15 min, test signals were developed. Sensitivity, specificity and other parameters of manufactured LFIA test strips were calculated and the results were compared with cefoxitin disc diffusion and PCR.
Statistical analysis: Statistical descriptive were performed by SPSS IBM SPSS Statistics 23. Using standard equations, the calculation of specificity, sensitivity, accuracy, and Positive Predictive Values (PPV) and negative predictive values (NPV) for each designed assay was conducted. Test and control bands intensity were measured using Image J 1.52i Software.
Results :
Characterization of the AuNPs and McAb-AuNPs conjugates: The absorption spectra of the synthesized nanoparticle with a UV-VS spectrophotometer was measured in the range of 400 to 600 nm and showed an absorption peak at a wavelength of 521 nm. The molar concentration of the synthesized nanoparticle was estimated and the molarity included 1.08×10-8 M. The size and shape of the AuNPs was examined by TEM. Also, the hydrodynamic size of the nanoparticles was determined by a DLS. The results of UV-VS, DLS and TEM are shown in figure 2. After binding the antibody to the AuNPs, the absorption wavelength also increased from 521 to 525 nm. The results of measuring the hydrodynamic diameter of AuNPs before and after conjugation antibodies to their surface with DLS revealed that the average size of AuNPs with dimensions of 14±2 nm has been changed to 15.22 nm.
Molecular identification of MRSA: Results of extracting the MRSA isolate genome are shown in figure 3 as well as results of the PCR of mecA gene for genotypic confirmation of MRSA strains.
LOD calculation: Analysis of the LFIA strips were visually showed by the appearance of positive T-line signal at 103 CFU/ml in the first strip and 104 CFU/ml in the second strip (Figure 4). The appearance of positive C-line signals was a confirmation of the correct function of the strip tests. Specificity of first strip was determined using strains other than Staphylococcus listed in the table 1 at concentration of 107 CFU/ml, which did not show signal appearance when any other species was tested.
For second strip, the specificity was confirmed using Methicillin-Susceptible Staphylococcus aureus (MSSA) (Figure 5).
Comparison of Sensitivity and specificity of LFIA assay with cefoxitin disc diffusion (phenotypic method) and PCR of mecA gene
Of the 52 MRSA isolates identified by cefoxitin diffusion disc (FOX30), 48 isolates were detected positively by LFIA and as resistant isolates, but 4 isolates were not detected by LFIA as MRSA. The sensitivity and specificity of the LFIA in the detection of MRSA isolates compared to cefoxitin diffusion disc (FOX30) is 92.30 and 97.36%, respectively. LFIA assay, PPV and Negative Predictive Value (NPV) were 94.11 and 96.52%, respectively. The results of the PCR method also show that of the 52 MRSA isolates identified by the PCR method, 48 isolates were positively identified by the LFIA test and as resistant isolates, but 4 isolates were not identified by the LFIA as MRSA. The sensitivity and specificity of LFIA in the detection of MRSA isolates compared to PCR method is 92.30 and 97.36%, respectively. These results were obtained using isolates suspended in 1X PBS and the clinical specimens were not used directly on the strip test. The results of our study have acceptable sensitivity and specificity compared to the PCR and cefoxitin disk diffusion (FOX30) methods (Table 2).
Discussion :
To reach a high sensitive and specific LFIA system some elements are considered of the most important factors. Defect in one, leads to failure and misleading results. Nanoparticle characteristics are the first or maybe the focal determiner to elevate the output quality. Here we tried to synthesize different size of AuNPs 14 and selected the average size of 14±2 nm as optimized nanoparticle. Its absorption spectra peak were consistent with the results of other articles 16,17. The shorter the absorption wavelength than 520 nm, the smaller the particle size 18,19. Also, the hydrodynamic size of the nanoparticles was determined by a DLS and the finding were similar to the research done by Yu Hll et al 20. After binding the antibody to the AuNPs, the absorption wavelength also increased from 521 to 525 nm, indicating the formation of bio conjugation. This results are quite similar to the results of related articles on the study of the McAb-AuNPs conjugate 21,22. The results of measuring the hydrodynamic diameter of AuNPs before and after conjugation antibodies to their surface with DLS revealed that the average size of AuNPs with dimensions of 14±2 nm has been changed to 15.22 nm. The results of these assays confirmed the conjugation between antibodies and AuNPs.
Genotypic (PCR based) and phenotypic method (antimicrobial susceptibility assessment), showed a 100% accordance for screening MRSA isolates. In this respect, our study was similar to that of Michael Otto, in which all MRSA strains isolated from different samples had the mecA gene 23. Analysis of the LFIA strips were visually showed by the appearance of positive T-line signal at 103 CFU/ml in the first strip and 104 CFU/ml and in the second strip. The most important factor in MRSA detection is the cut-off detection point, has been reported from 105 to 106 CFU/ml in the few articles 9,12,13. In this study, we were able to improve the cut-off to 104 CFU/ml by optimizing the impressive parameters and choosing the desirable target and detector elements. The first strip was used to identify S. aureus isolates. The strategy used in this strip to amplify the signal and increase the sensitivity of the LFIA test is to use a two-label system (two antibody-AuNPs conjugate pads). The intensity of the signal in the test line as well as the sensitivity increased due to the increase in the number of nanoparticles relative to the sample, so that the detection limit of S. aureus isolates in the first strip reached 103 CFU/ml.
Optimal size selection of an AuNPs in LFIA is of vital milestone. In the present study, the best results were obtained when both conjugated nanoparticle were the same in size in the first strip, which was quietly adopted with those of Shen et al 24. But the study by Choi et al has showed opposed results. They used smaller nanoparticles for the first conjugate solution and larger nanoparticles for the second conjugate 25. Sensitivity and specificity of the LFIA in the detection of MRSA isolates compared to cefoxitin diffusion disc (FOX30) and PCR is 92.30 and 97.36%, respectively. Additionally, 48 isolates were positively identified by the LFIA test and as resistant isolates, but 4 isolates were not identified by the LFIA as MRSA. The results of our study presented acceptable sensitivity and specificity compared to the PCR and cefoxitin disk diffusion (FOX30) methods.
Hidehito Matsui et al, in 2011 developed a monoclonal antibody against PBP2a. In their study, 62 clinical isolates of S. aureus and 53 isolates of CNS were tested with LFIA, PCR and latex agglutination test. PCR detected only 37 mecA positive isolates within 62 clinical isolates of S. aureus. LFIA and latex agglutination had similar results to PCR. In this study, the accuracy of PCR and LFIA detection was the same. And similar to our study, these results were obtained on clinical isolates not clinical specimens. In this study, despite cut-off of 105 CFU/ml, LFIA method accuracy in MRSA detection was reported to be 100% 9. Surasa Wiriyachaiporn et al diagnosed S. aureus in respiratory sample using the LFIA. Despite the use of monoclonal antibodies specific for peptidoglycan, they reported the 106 CFU/ml detection limit for S. aureus isolates. However they do not detect MRSA 12.
Conclusion :
In this study, a high-sensitive LFIA strips with detection limit of 104 CFU/ml was developed. The results of the present study show that the developed LFIA strips have acceptable sensitivity and specificity for detection of MRSA. Compared with conventional bacterial methods (e.g., FOX30 and PCR), LFIA can detect MRSA and S. aureus quickly and directly from the visual signal within 25 min with the naked eye and it is suitable for routine tests in laboratories.
Acknowledgement :
The authors are thankful to the personnel of Department of Pathobiology, School of Public Health, TUMS. We also appreciate Tehran University of Medical Sciences Research Committee for their financial support of this study (grant number no. 37134).
Conflict of Interest :
The authors declare no conflict of interests.
Funding: This study was supported by Tehran University of Medical Sciences Research Committee under grant number no. 37134.
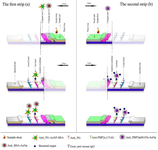
Figure 1. Schematic of multiplex LFIA system based on a dual AuNPs conjugate. a) Peptidoglycan strip (S. aureus specific strip), b) PBP2a strip (MRSA specific strip).
|

Figure 2. A) Characterization of the bare AuNPs (14±2 nm) and conjugated AuNPs whit visible spectra of AuNPs B) TEM of AuNPs and C1) Dynamic light scattering metrics (DLS spectrum) of bare AuNP (14 ±2 nm) and c2) DLS spectrum conjugated AuNPs.
|
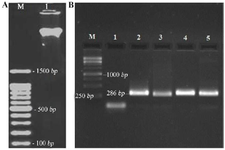
Figure 3. Molecular identification of MRSA. A) Electrophoresis results of MRSA genome extraction, Lane M: 100 bp molecular size marker. Lane 1: Genomic DNA extracted. B) PCR products of mecA gene, Lane M: 1 kb molecular size marker. Lane 1: Negative Control for mecA (MSSA). Lane 2: Positive Control for mecA (286 bp). Lane 3-5: positive isolate for mecA gene.
|
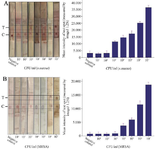
Figure 4. Limit of detection of LFIA strips. A) Peptidoglycan strip (S. aureus specific strip). B) PBP2a strip (MRSA specific strip). Imaging analysis of the LFIA strips showed that the first significant signal in the detection of S. aureus isolates in the first step (a) was at 103 CFU/ml and the first significant signal in the detection of MRSA isolates in the second step (b) was at 104 CFU/ml.
|
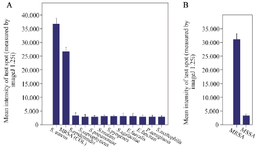
Figure 5. Results of specificity of the LFIA strip. Results showed that A) the first strip had no signals for strains other than Staphylococcus, B) the second strip also had no signal for MSSA.
|
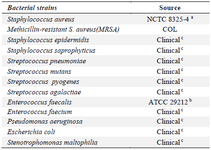
Table 1. Bacterial strains and clinical isolates used in this study
a: National collection of type cultures. b: American type culture collection.
c: Diagnostic laboratories of Tehran University.
|

Table 2. Results of sensitivity and specificity LFIA compared with FOX30/PCR
Diagnostic sensitivity= true positives (a)/ true positives (a) + false negatives (b).
Diagnostic specificity= true negatives (d)/ true negatives (d) + false positives (c).
Positive predictive value (PPV) = true positives (a)/ true positives (a) + false positives (c).
Negative predictive value (NPV) = true negatives (d)/ true negatives (d) + false negatives (b).
|
|