Cellular Therapy: The Hope for Covid-19
-
Nobari, Sima
-
Research Center for Molecular Medicine, School of Medicine, Hamadan University of Medical Sciences, Hamadan, Iran
-
Rezvan, Motahareh
-
Department of Photo Healing and Regeneration, Medical Laser Research Center, Yara Institute, ACECR, Tehran, Iran
-
Dashtestani, Fariba
-
Institute of Biochemistry and Biophysics, University of Tehran, Tehran, Iran
-
Gangi, Mahdieh
-
Department of Biology, Payame Noor University, Taft, Iran
-
 Keshmiri Neghab, Hoda
Department of Photo Healing and Regeneration, Medical Laser Research Center, Yara Institute, ACECR, hodakeshmiri@ut.ac.ir
Keshmiri Neghab, Hoda
Department of Photo Healing and Regeneration, Medical Laser Research Center, Yara Institute, ACECR, hodakeshmiri@ut.ac.ir
-
Department of Photo Healing and Regeneration, Medical Laser Research Center, Yara Institute, ACECR, Tehran, Iran
Abstract: Coronaviruses (CoVs) are a group of very diverse viruses that cause a broad spectrum of diseases from mild to severe enteric, respiratory, systemic diseases, and common cold or pneumonia among humans and animals. This virus is associated with Middle East Respiratory Syndrome (MERS), Severe Acute Respiratory Syndrome (SARS), and lung disease that lead to Acute Respiratory Distress Syndrome (ARDS). In December 2019, researchers identified a novel coronavirus type, called Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV2), which was associated with symptoms of high fever, dry cough, headache, diarrhea, and reduction of White Blood Cells (WBC). Coronavirus-associated acute respiratory disease was named Coronavirus Disease 19 (COVID-19). No proven treatment has been discovered for COVID-19 so far, but researchers are trying to find the best effective way to treat this disease. Therefore, therapeutic strategies that facilitate the recovery of COVID-19 patients and reduce life-threatening complications are urgently needed now. Today, Mesenchymal Stem Cells (MSCs) and their secretion are utilized as one of the most applied tools to treat various diseases such as inflammation and cancer. MSC-derived vesicles are rich in various growth factors, cytokines, and interleukins that are produced and secreted under different physiological or pathological conditions. These vesicles were considered a suitable and effective tool in regeneration medicine because of their high power in repairing damaged tissues and modulating immune responses. Recently, evidence has shown MSC-derived vesicles through reduced expression of pro-inflammatory cytokines could improve damaged tissues in COVID-19 patients. In addition to MSCs and MSC-derived exosomes, Natural Killer (NK) cells, T cells, and platelet lysates were used against viral infection. In this review, we tried to provide an overview of MSC secretion and immune cells for COVID-19 therapy.
Introduction :
Stem cells are undifferentiated cells that can differentiate into mature specialized cells. Some of the stem cells such as Mesenchymal Stem Cells (MSCs), Hematopoietic Stem Cells (HSCs), and skeletal muscle stem cells play a crucial role in cell therapy 1.
Recently, cell therapy has become a beneficial tool for disease treatment as well as a strategy for replacing, repairing, and enhancing the biological function of damaged tissue. The use of cell therapy in treatment of diseases such as cancer, autoimmune disease, joint in-juries, and infectious diseases has been proven 2.
The COVID-19 disease is caused by a positive-stranded RNA virus named SARS-CoV2. This virus binds to host Angiotensin-Converting Enzyme 2 receptors (ACE2) through the viral spike protein, leading to excessive stimulation of immune system and consequently damaging body organs 3-5. ACE2 by converting Angiotensin II (AngII) increases the levels of Tumor Necrosis Factor-alpha (TNF-α) and Interleukin 6 (IL-6) in the cells that lead to upregulating Nuclear Factor kappa B (NF-κb) and activating the inflammasome 6. ACE2 receptors are present in many tissues including the lungs, heart, blood vessels, kidneys, liver, and gastrointestinal tract, and also are abundant in human Alveolar Type II cells (AT2) 7.
The replication of SARS-CoV2 in target cells in duces the release of large amounts of inflammatory factors from host immune cells, causing a cytokine storm. This exaggerated immune response is responsible for different complications in the host and eventually leads to multiple organ failure 8. Numerous research-es have shown higher plasma levels of Granulocyte Colony-Stimulating Factor (GCSF) and TNF-α in severe COVID-19 patients, and on the other hand, increasing GCSF and TNF-α levels induce cytokine storm and lead to Acute Respiratory Distress Syndrome (ARDS) 9. So, suppressing immunological response and regeneration of the lung tissue can be an effective treatment modality for COVID-19 patients 10.
Researchers are trying to recommend the best treatment for COVID-19 disease. Several studies show that the use of MSCs and their secretion can treat coronavirus-induced pneumonia 11. MSCs therapy offers a promising approach for reducing the harmful effects in COVID-19 patients. This therapeutic approach reduces the expression of pro-inflammatory cytokines as well as repairs damaged tissues in COVID-19 patients 9. Except for MSCs, immune system cells, including Natural Killer (NK) cells and platelet lyses play an important role in the body's defense against disease 12. This review has been organized to put forward the positive arguments and implications in support of cell therapy as a necessary approach for treating COVID-19 patients (Figure 1).
Role of mesenchymal stem cells (MSCs) in respiratory
virus-induced lung injury
Despite the development of stem cell-based therapy, important limitations such as immunogenicity, limited cell source, and ethical issue have not been solved yet. Among these, MSCs have attracted attention due to: easily accessible with a high proliferation rate, low invasive procedures, and being free of ethical issues 13. MSCs are spindle multipotent stromal cells. These stem cells are derived from different adult tissue and express special cell surface markers (positive for CD73, CD90, and CD105; negative for CD45, CD34, CD14 or CD11b, CD79 or CD19, and HLA-DR). MSCs have self-renewal capability and are differentiated into several cell-types 14. MSCs as non-hemato-poietic cells are normally found in the bone marrow and make up about 0.001-0.01% of all bone marrow nucleated cells. MSCs are easily accessible and derived from multiple tissues such as bone marrow and adipose tissues, umbilical cord, dental pulp, menstrual blood, buccal fat pad, etc. These stem cells demonstrate similar phenotypes but have different functions and variable markers in each tissue 15. MSCs can easily expand to clinical volume in a suitable period and can be stored for repetitive therapeutic usage 11.
The immunoregulatory properties and the tissue reparative ability of MSCs are relied on express immunosuppressive molecules as well as the secretion of in-flammatory cytokines, and various growth factors that induce tissue repair and maintain immune homeostasis 16,17. Furthermore, MSCs are capable of activating other resident stem cells to participate in the healing process and can stimulate neo-angiogenesis, tissue repair, and cell survival in surrounding tissues 18. MSCs could retain Dendritic Cells (DCs) in an immature state by suppressing pro-inflammatory cytokine secretion and via inhibiting the expression of Major Histocompatibility Complex (MHC) class II, CD1-α, CD40, CD80, and CD86 markers 19. MSCs through increasing secretion soluble factors, such as TGF-β and Prostaglandin E2 (PGE2) can inhibit the function of DCs, NK cells, and macrophages 20. These stem cells decrease IL-2- or IL-15-driven NK cell proliferation 21.
MSCs have been already known to inhibit T-cell proliferation in adaptive immune responses 22. Besides, they can decrease B-cell proliferation via cell-cell contact and secret growth factors 23. MSCs induce IL-10-secreting macrophages in both in vitro and in vivo researches 24. When MSCs have entered the microenvironment of damaged tissues, several different cytokines such as TNF-α, IL-1, IFN-γ, toxins of infectious agents, viruses, and hypoxia can trigger the secretion of critical soluble growth factors by MSCs, including Epidermal Growth Factor (EGF), Fibroblast Growth Factor (FGF), Insulin Growth Factor-1 (IGF-1), Transforming Growth Factor-β (TGF-β), Vascular Endothelial Growth Factor (VEGF), Hepatocyte Growth Factor (HGF), Platelet-Derived Growth Factor (PDGF), Angiopoietin-1 (Ang-1), Keratinocyte Growth Factor (KGF) and Stromal cell-Derived Factor-1 (SDF-1). These growth factors increase the development of fibroblasts, integrity of endothelial cells and promote angiogenesis through regulating endothelial cell proliferation and extracellular matrix production and subsequently tissue repair accrue 25-28. These cells play their role by direct cell to cell contact and secretion of growth factors 7. MSCs can release extracellular vesicles containing numerous mRNAs, regulatory miRNAs, multiple bioactive proteins and compounds, and the production and secretion of a large number of regulatory substances rather than MSC direct differentiation and cell replacement 29,30.
The basic therapeutic effects of MSCs are now attributed to the stimulation of several endogenous repair processes in injured tissues in vivo by secreted factors as well as the modulation of an immune response, which translates into a positive outcome of MSC-based therapies 31,32. Therefore, MSCs have been mentioned as an effective cell source for cell therapy approach 33.
A lot of clinical trials using MSCs in different aspects have been down 34-37. MSC-based therapies are superior to other treatments due to their source potential, high proliferation, low invasiveness, and being free of ethical issues 13. Additionally, MSCs based immunomodulation treatment has been one of the promising tools in various diseases and several clinical trials have begun in this regard 38. MSC administration can significantly reduce lung injury in respiratory viruses such as influenza strains H5N1 and H9N2 39.
After the entry of COVID-19 into the body an exaggerated immune response is induced through the production of large amounts of inflammatory agents, including cytokines, chemokines, and immune cell responses. Evidence has shown MSCs in lung cells can release different soluble mediators including cytokines, antimicrobial peptides, angiogenic factors, and extracellular vesicles 40-42. In systemic administration MSCs lodge in the pulmonary vascular. MSCs clearance from capillary endothelial cells via apoptosis and phagocytosis by resident inflammatory, immune cells, and macrophages is around 24-48 hr 43,44.
Numerous reports strongly focus on MSC paracrine properties in ARDS patients, as well as, introducing MSCs as a therapeutic approach for use in the treatment of COVID-19 45. Leng et al performed a clinical trial study (ChiCTR2000029990) using human Umbilical Cord-Derived MSCs (hUCMSCs) in seven COVID-19 patients for 14 days and then observed that the population of CD14+, CD11c+, CD11bMid and regDCs mark- edly increased and levels of TNF-α decreased, while levels of IL-10 increased in the MSC treatment group compared to the control group 9. Recent experiments on rat lungs have shown that MSCs can be modulate inflammatory reactions that lead to acute lung injury and pulmonary fibrosis 46. MSCs, inhibit tissue fibrosis and stimulate angiogenesis by producing anti-fibrotic and angiogenic factors 47. The summary of MSCs mechanisms of therapy is given in table 1.
Despite MSCs attracting a lot of attention in regeneration medicine, it seems MSCs therapies are complicated and their outcomes should be followed-up in long term. Also during MSCs transplantation potential risks such as pro-tumorigenic, immune response, disturbed differentiation capacities, differentiation into desirable tissue, short survival after transplantation, unspecial optimal dose and route of cell administration should be considered 2,48,49. Some evidences demonstrated that the quality of MSCs in cell therapy depends on the age, genetic traits, and medical history of the donor 50,51. Another complicated issue in the use of MSCs in autologous transplantation is how to expand MSCs from elderly patients to obtain a suitable number of interest cells. Also, it is difficult to isolate an effective population of MSCs from patients with some diseases because cells isolated from the patient may be affected by the disease 52.
Extracellular vesicles derived- MSCs
Extracellular Vesicles (EVs) are small circulating membrane-enclosed entities shed from cell surfaces of variant cell types in response to cell conditions 53. EVs measure 0.01-4 μm and are generated by various processes. The secretion of EVs depend on chemical and mechanical stimuli. In addition gamma-ray, calcium ionosphere, heparinase, statins, hypoxia, and acidic condition can also increase the secretion of EVs 54,55. EVs are divided into three categories based on size and cell sources such as 1) Ectosomes or Microvesicles (MVs) (200~1000 nm) that originate from the plasma membrane, 2) Exosomes (40~100 nm) are composed of the germination of intracellular secondary endosomes that are secreted out of the cell after fusion with the cell membrane, and 3) apoptotic bodies (50~500 nm) are released from a cell that has been in apoptosis 56.
Stem cell secretome induces the neighboring cells to modulate various downstream pathways, including im-munomodulation, suppression of apoptosis, prevention of fibrosis, and remodeling of the injured tissues 57. Stem cells release these secretomes by common secretory mechanisms. When secretomes are injected into the patients, the near cells absorb the molecules by paracrine signaling 58. MSCs secretome can repair damaged tissue, reduce tissue fibrosis, express antiviral miRNA genes to improve the function of immunity by directly affecting viral replication 59. As mentioned MSCs exosomes are types of EVs that have shown significant potential in recent decades 60. Indeed, researchers found that plasma EVs can affect directly patients' host cells and modulate immune systems 61. MSC-Exos can inhibit T CD8+ and T CD4+ via inhibiting T cells expression IL-17 and induced IL-10-expressing 62. MSC-Exos could suppress differentiation of T CD8+ and T CD4+ cells via releasing TGFβ and inhibit inflammation in vivo model 63. MSCs exosomes can repair the muscles by the process of myogenesis and angiogenesis with function depending on miR-494 in the exosomes 64.
MSCs exosomes, by inducing gene expression in the target cell can regenerate bone marrow stroma and lead to an increase in the function of angiogenesis in target cells. EVs are one of the ideal carriers for drug or gene delivery to the target tissue because they are rich in various growth factors 65. Evidence shows direct use of MSCs should increase the risk of rejection but EVs are successful in transplantation with no rejection. EVs can be a good alternative for MSCs in the treatment and repair of tissues 66.
Faisal A. Alzahrani et al, showed ExoFlo™, a bm-MSC-derived exosome agent, significantly improved neutrophilia and lymphopenia including increased CD3+, CD4+, and T CD8+ lymphocytes in addition to reducing the severity of diseases 67. MSCs mediate these kinds of effects by direct contact, where it releases the regulatory cytokines, such as Interferon-gamma (IFN-γ), indoleamine 2,3-dioxygenase, TGFβ, IL-10, and PGE2 68. Moreover, MSCs can hinder the proliferation and/or functions of the CD4+ Th1 and TH17 cells, T CD8+ lymphocytes, and the NK cells, mainly by secreting soluble factors and vesicles, such as TGFβ1 and HGF 69. MSCs secretome has a direct and indirect effect on modulating immune responses. MSC injection in Acute Lung Injury (ALI) and ARDS preclinical models resulted in improved lung tissue recovery. MSCs secretome increase the level of anti-inflammato-ry molecules such as IL-10 while decreasing inflammatory cytokines, including TNF-α, macrophage inflammatory protein 2 (MIP-2), IL-1β, TGF-β, VEGF, IL-6, IFN-γ, Myeloperoxidase (MPO), Regulated upon Activation Normal T Cell Expressed and Presumably Secreted (RANTES), and Nitric Oxide Synthases (NOS) levels 70,71. The list of extracellular vesicles derived-MSCs used in COVID19 clinical trial is given in table 2.
It should be noted that despite the therapeutic effectiveness of MSC-EV in preclinical studies, the use of EV in clinical treatments has several critical issues such as (i) isolation methods and large-scale-product-ion, (ii) rapid and accurate characterization method, (iii) precise content characterization of the cargo, (iv) pharmacokinetics, targeting and transfer mechanisms of EV to the accurate sites, and (v) safety profiles to access the best clinical dosage and possible toxicities upon repeated administration 72.
Natural killer cell (NK) based therapy in Covid-19
NK cells are the first responder's defense system against viral infections because of their role in cytolysis and chemokine releases. The NK cells are cytotoxic lymphocytes that contain 10-15% of total peripheral blood leukocytes in humans. These cells play a crucial role in linking innate and adaptive immune system activity 82. They are present in the peritoneal lymph node, thymus, spleen, liver, peritoneal hollow area, and gravid uterus. Their function is well mediated by the ligand-receptor interaction. NK cells' role in tumorigenic, asthma, autoimmune diseases, and HIV-AIDS have been well defined 83. The interaction of NK cells in viral infections amplifies their potential role in showing the innate immune response; increasing the IFN-γ release; direct cytolysis of target cells and viral replication inhibition 84. After attachment of virus-infected cells to the Natural Killer Group 2D (NKG2D) receptor, major activating NK cell receptor NKG2D, NK cells are activated and produce anti-microbial and immunoregulatory cytokines and ultimately kill the infected cells. All NKG2 receptor families are type II transmembrane glycoproteins. Also activating receptor NKG2D (CD314) is expressed in NK. This finding describes the rational function of the NK cell in COVID-19 85. In COVID-19 patients’ number of NK cells are decreased which is associated with development and severity of COVID-19 86.
The most common symptoms of coronavirus infections include significant morbidity and mortality related to pulmonary lesions and acute respiratory syndrome due to an exaggerated immune response, where NK cells are a critical mediator. Along with their effective antiviral function, NK cells have also been related to immunopathology in infections like Respiratory Syncytial Virus (RSV), influenza, and hepatitis B 87,88.
The natural immunity response is triggered by molecular structures -called Pathogen-Associated Molecular Patterns (PAMPs) that are recognized by Pathogen Recognition Receptors (PRRs). In respiratory RNA viruses, epithelial cells, which express certain PRRs, are infected and have a primary role in the first line of defense. Toll-Like Receptor (TLR3), TLR7, TLR8, Melanoma Differentiation-associated protein 5 (MD-5), and Retinoic acid-inducible gene I (RIGI) are PRRs predominantly expressed by immune and non-immune cells against RNA respiratory viruses, like coronaviruses. Following identifying PRRs, production type I interferons (IFNs), (IFN-a/b) and other components involved in the inflammatory response, are the main stage in antiviral responses.
Also, decreased NK cell levels have been inversely associated with the severity of COVID-19 patients. One of the possible mechanisms by which NK cells can also emerge as hyporesponsive in fighting coronavirus is interference with type I IFN pathways 89.
IFNs type I is released by both immune and non-immune cells and besides stimulating interstice antiviral defense mechanisms, are critical to natural and adaptive lymphocytes, such as NK cells. In patients with Natural Killer cell Deficiency (NKD) and immune-compromised who are highly sensitive to viral infections, the importance of NK cells as a first-line antiviral responder is highlighted. Some studies proposed that neutrophils and inflammatory monocyte-macrophages accumulate in the lungs in acute coronavirus infections such as, Severe Acute Respiratory Syndrome (SARS) and Middle East Respiratory Syndrome (MERS) and release the chemokines and cytokines that result in NK cell activation and migration 90.
Additionally, adoptive NK cells and CAR-NK cells are candidates for cell-based therapy as an alternative treatment of COVID-19 patients. In a new report, Novocellbio has confirmed that its autologous NK cell treatment agent Novo-NK, an autologous natural killer cell treatment, is effective in killing SARS-CoV-2 in a cellular and preclinical study. Recently, five clinical trials have been registered at the Clinical level based on using of Peripheral Blood Mononuclear Cell (PBMC) derived NK cells in COVID-19 patients, although, there have been no published clinical reports yet. In one of these trials (Identifier: NCT04344548), patients were treated with three different injections of NK cells 48 hr apart with 1, 10, and 20, 106 cells/kg body weight, but the protocols are unknown and clinical trials are in phases 1 and 2 91.
T cell-based therapy in COVID-19
The T lymphocytes are one of the important blood cells which play a critical role in adaptive immune response. T lymphocytes driven from hematopoietic stem cells in bone marrow, then migrate to the thymus gland for maturation. T cell exhaustion is a broad term that was known as the response of T cells to chronic antigen stimulation, bacterial, and parasitic infections as well as human cancers 92. Lately, two backgrounds have shed light on the comprehension of T cell exhaustion. Firstly, both intrinsic negative regulatory pathways (such as PD-1) and extrinsic regulatory pathways (along with immunoregulatory cytokines) have the main role in T cell exhaustion 93. T cells play a main role in development of COVID-19 patients. Transient lymphopenia is a typical characteristic in many respiratory viral infections, including influenza A H3N2 virus, human rhinovirus, or respiratory syncytial virus. Lymphopenia is related to the severity of the COVID-19 and some studies have shown reduction of CD3+, CD4+, CD8+ T lymphocyte, B cells, and NK cells in COVID-19 patients. Similarly, the most recent RNA-seq upper respiratory tract reported that the participation of cytotoxic T lymphocytes in patients with COVID-19 lymphocytes significantly decreased compared to mild disease. Several kinds of research demonstrated lymphopenia as an eminent feature of the SARS-CoV-2 infection 94. Thus, finding out how lymphopenia could influence T cell hyperactivation in patients with COVID-19 is a crucial aim in therapeutic approaches. There are some studies that strongly suggest the formation of immunological memory of T cells following improvement from SARS-CoV-2. This evidence highlights the important role of T cells in the diagnosis and treatment of severe patients 95.
Platelet lysate based therapy in COVID-19
Human blood is known as a source of many therapeutic agents which can be classified into cellular and protein-based blood products. The cellular blood products include buffy coat derived, red blood cells, and apheresis platelets, whereas protein-based products are cryoprecipitate, fresh frozen plasma, and plasma fractionation products. Platelet lysate is an acellular product consisting of regenerative molecules released from a cluster of platelets and considered as a regenerative immunomodulatory agent to combat COVID-19 96. The critical role of platelets in wound regeneration and tissue reconstruction is feasible by the use of human platelet derivatives products in regenerative medicine 97.
Platelet lysate decreases immune reaction with the introduction of growth factors, cytokines, and proteins. Cytokines and factors in platelet granules will be released naturally by stimulation of thrombin and coagulation or artificially by sonication, chemical agents or freeze/thaw-mediated platelet lysis. Platelet lysate can be used in nebulized form for such acute respiratory distress conditions in COVID-19 elderly patients. Evidence has shown the effect of the platelet in COVID-19-associated pathophysiology.
Furthermore, platelet lysate can be used for treating problems such as chronic obstructive pulmonary disease, acute lung injuries, and asthma. In this manner, as adjuvant therapy for the treatment of COVID-19 associated acute respiratory syndrome, it can be useful because it triggers inflammatory processes 98. Platelets release proinflammatory cytokines that promote inflammatory immunomodulation. There is a multiplex interplay between the intrinsic and adaptive immunological components of platelets in the clearance of pathogens. The role of platelets in defending against viral infections has been reported by research and requires accurate interaction of the receptor-ligand. The role of platelets and their interaction in viral infection has been characterized. Furthermore, it can increase the oxygen saturation range for patients with COVID-19 99.
Platelets are stimulated either by direct or indirect stimuli and undergo degranulation. In addition, it releases numerous growth factors and biomolecules that are active in mediating host defensive mechanisms. New chemokines that also exert direct antimicrobial activity have been termed kinocidins. Platelet factor 4 (PF-4/CXCL-four) has been listed as the most effective minocycline. CCL-five has been reported to be active in the viral affected lung, and this has been shown in viral infection of influenza A in mice, where it provides (anti-apoptotic) signalling for macrophages. Like-wise, platelet lysate can grow to be a key participant in viral lysis 100.
The platelet lysate processing produces a rich material content of growth factors with a limited volume of white cell antigens. The platelet lysate will provide a beacon of desire in cell regeneration by stimulating cell proliferation and angiogenesis with the supply of supraphysiologic doses of platelet factors. This trait can be further analyzed to act as an adjuvant in patients with COVID-19.
Platelets have bioactive molecules that, once platelets are used in the form of platelet-rich plasma, cause the quiescent cells to perform the target effects. The next application of platelet technology is platelet lysate obtained from the ruptured platelet membrane, and these acquired bioactive molecules are then based on using lyophilization to manufacture powdered form; the mission here is the lyophilization and immunogenicity process. The advantage of platelet lysate is the quantification of bioactive molecules present in it. However, in multiple diseases, it is an expanded way to verify dosage and control strategies 101.
Conclusion :
There is no specific treatment for COVID-19 now, and the recent treatment methods for COVID-19 are confined to the previous therapeutic techniques for similar viruses and previous generations of coronaviruses, including SARS- and MERS-CoVs. Such a situation calls attention from various medical fields including stem cell therapy and immune-modulatory methods. COVID-19 can damage various systems at the same time, including the central nervous system, the gastrointestinal system, and the respiratory system, and this will depend on its profound effects on the immune system. The improving features of MSCs, including their regenerative properties and ability to differentiate into diverse cell lineages, have generated considerable interest among researchers whose work has offered intriguing perspectives on cell-based therapies for various diseases. Immunomodulatory and anti-inflammatory properties of MSCs in the treatment of respiratory diseases were confirmed. However, the cost-effectiveness and speed of therapeutic preparation are the capable discussed issue for MSC-based therapy for COVID-19. Besides, the clinical use of MSCs therapy to treat COVID-19 is still some time away, but there are some promising reports to utilize it in the clinical trial. Stem cell therapy and especially MSCs maybe be one of the most ideal therapeutics or a combination of treatments to treat COVID-19 patients. However, scientists are trying incessantly to develop a vaccine for COVID-19, as well as therapeutics to treat this disease. The immunomodulatory effects of MSCs, which may assist in inhibiting cytokine storm and lung inflammation, are of particular interest for COVID-19 therapy. Besides, the potentials of NK cells to exert cytotoxic effects on infected cells and induce interferon production perhaps make NK cells a promising candidate for COVID-19 cell therapy. Cell-derived exosomes can be used as transfers for various macromolecules, which can modulate immune cells and signaling pathways, and therefore, become interesting for COV-ID-19 therapy. Finally, induced Pluripotent Stem Cells (iPSCs) can help the development of a personalized approach to COVID-19 therapy.
Generally, it has been shown cell-based therapies may be applicable in specific pathogenesis of COVID-19. However, there are some limitations to cell therapies that must be considered including immune rejection, dosing of the cells, number of injections, cell delivery approaches, cell survival/retention, and age of patients 102. The appropriate route of administration is very important in effectiveness of cell therapies. Furthermore, multiple strategies are being addressed to boost cell viability, amend functional features of individual cells, and to elongate cell retention. For instance, integrating pharmacotherapy and cell therapy, genetic modification and preconditioning cells are strategies which are being studied to enhance cell therapy efficiency. Another alternative strategy is the use of a combination of several cell types. For example, epithelial cells and pulmonary endothelial cells have been investigated for their synergistic effect with MSCs 103. In sum, the present comprehension of cell based therapy and the finding strategies will certainly progress cell-based therapy and treat patients with Covid-19.
Conflict of Interest :
The authors have no financial or non-financial conflicts of interest to declare.
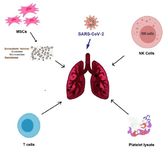
Figure 1. Schematic representation of cell-based therapeutic appro-aches against covid-19.
|
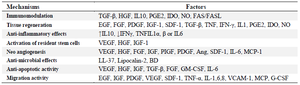
Table 1. The summary of MSCs mechanisms of therapy
|
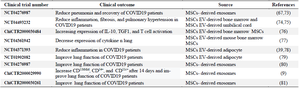
Table 2. MSC-EV in the treatment of COVID-19
|
|