Investigation of Integron-Associated Resistance Gene Cassettes in Urinary Isolates of Klebsiella pneumoniae in Yasuj, Southwestern Iran during 2015-2016
-
Jahanbin, Fariba
-
Department of Basic Sciences, Islamic Azad University, Yasuj Branch, Yasuj, Iran
-
Marashifard, Masoud
-
Treatment Management of Social Security Organization of Kohgiluyeh and Boyer-Ahmad Province, Yasuj, Iran
-
Jamshidi, Sanaz
-
Department of Basic Sciences, Islamic Azad University, Yasuj Branch, Yasuj, Iran
-
Zamanzadeh, Maryam
-
Department of Basic Sciences, Islamic Azad University, Yasuj Branch, Yasuj, Iran
-
Dehshiri, Masumeh
-
Cellular and Molecular Research Center, Yasuj University of Medical Sciences, Yasuj, Iran
-
 Khoramrooz, Seyed Sajjad
Medicinal Plants Research Center, Yasuj University of Medical Sciences, Yasuj, Iran, Tel/Fax: +98 743 323 5153; E-mail: Khoramrooz@gmail.com, masoud.marashifard@gmail.com
Khoramrooz, Seyed Sajjad
Medicinal Plants Research Center, Yasuj University of Medical Sciences, Yasuj, Iran, Tel/Fax: +98 743 323 5153; E-mail: Khoramrooz@gmail.com, masoud.marashifard@gmail.com
-
Medicinal Plants Research Center, Yasuj University of Medical Sciences, Yasuj, Iran
-
Department of Microbiology, Faculty of Medicine, Yasuj University of Medical Sciences, Yasuj, Iran
Abstract: Background: Growing antibiotic resistance among urinary opportunistic pathogens such as Klebsiella pneumoniae (K. pneumonia) has created a worrisome condition in the treatment of the Urinary Tract Infections (UTIs) in recent years. Integrons play a significant role in the dissemination of antibiotic resistance genes. The present study was conducted to investigate class 1-3 integrons and the corresponding resistance gene cassettes in urinary K. pneumoniae isolates.
Methods: In this study, from December 2015 to September 2016, a total of 196 K. pneumoniae isolates were collected from the patients with UTI referred to medical diagnostic laboratories in Yasouj, Southwestern Iran. Antibiotic susceptibility patterns of isolates were determined using 12 antibiotics by the disc diffusion method. Polymerase Chain Reaction (PCR) was used for detection of integron genes (intI1, intI2, and intI3). The variable regions of integrons were amplified by PCR and sequenced to identify the corresponding gene cassettes.
Results: Thirty-nine different antibiotic resistance profiles were observed among K. pneumoniae isolates. Only 12.2% of K. pneumoniae isolates were found to harbor the intI1 gene. While 17 (60.7%) out of 28 Multidrug Resistance (MDR) K. pneumoniae isolates carried the intI1 gene, only 4.2% of non-MDR isolates harbored intI1 gene. Totally 7 different gene cassette arrays were found in the intI1 gene of K. pneumoniae isolates. The aadA1 was the most prominent gene cassette. Also, high frequency of dfrA containing gene cassettes was observed.
Conclusion: Continuous monitoring and characterization of integrons and their associated gene cassettes could be helpful in controlling the rising rate of antibiotic resistance.
Introduction :
Urinary Tract Infection (UTI) is one of the most prominent infectious diseases in both community and healthcare setting. Escherichia coli (E. coli) is considered as the most prominent uropathogen which accounts for 75-90% of all UTIs in both inpatients and outpatients followed by Klebsiella pneumoniae (K. pneumonia) 1,2. The increasing resistance rate to antibiotics among UTI causing organisms makes the empiric treatment of these infections very challenging 3. Increasing antibiotic usage, as well as horizontal transfer of antibiotic resistance genes located on various types of mobile DNA elements as plasmids, transposons and gene cassettes in integron, has facilitated the development of Multidrug Resistance (MDR) in Enterobacteriaceae family 4. Integrons are genetic elements that play a significant role in the transmission of multidrug resistance genes in several gram-negative bacteria 5. An integron is structurally composed of three genetic elements; integrase gene (intI) which is responsible for site specific recombination of mobile gene cassettes, attachment site (attI) and the promoter (Pc) 6. Gene cassettes are discrete genetic elements consisting of the single Open Reading Frame (ORF) and a recombination site (a 59 bp element) known as the attC site 7. The integrase mediates the integration of circular gene cassettes between attI and attC sites 8. Integrons are classified into several classes, based on the amino acid sequence homology of intI gene, among them class 1, 2 and 3 are usually recovered from clinical isolates 9,10. Class1 integron has been reported in different studies as the most frequent class identified in clinical isolates 9. Class1 integron contains variable regions of gene cassettes that are sometimes absent in the structure of integron, flanked by two highly conserved regions; 5′-conserved segment (5′-CS) and 3′-conserved segment (3′-CS) 6. Gene cassettes are promotorless variable regions of integrons, which encode antibiotic resistance phenotype, located between attC and attI region. Their expression depends on the integron promoter (Pc) which relies on the 5′-CS in the case of class1 integron. Different arrays of cassettes have been reported and most arrays had two or three gene cassettes 11.
At least 130 different gene cassettes including diverse resistance genes have been identified, mainly conferring resistance to different classes of antibiotics including aminoglycosides, β-lactams, chloramphenicol, trimethoprim, erythromycin, and rifampicin 11,12. The most prominent gene cassettes within class 1 integron among Enterobacteriaceae family are aadA and dfrA, which confer resistance to streptomycin and trimethoprim, respectively 13. Although some studies have reported the prevalence of class 1-3 integrons and gene cassettes in E. coli, Pseudomonas aeruginosa and Acinetobacter baumannii isolates in Iran 14-17, there are limited data regarding the distribution of gene cassettes in K. pneumoniae isolates from Iran 18. Therefore, in the present study, an attempt was made to investigate the gene cassettes in addition to the prevalence of class 1-3 integrons and antibiotic resistance patterns in a series of clinical isolates of K. pneumoniae in Yasuj, Southwestern Iran.
Materials and Methods :
Bacterial isolation: From December 2015 to September 2016, 196 non-repetitive isolates of K. pneumoniae were collected from the urine samples of patients with UTI who were referred to medical diagnostic laboratories in Yasouj, Southwestern Iran. Each urine sample was cultured on EMB and Blood Agar (Merck, Germany) and incubated at 37°C for 24 hr. Using conventional biochemical tests on culture media such as Methyl red-Voges Proskauer (MR-VP), Triple Suger İron (TSI), Sulfide İndole Motility (SIM), Simmons Citrate, and Urea Agar (Merck, Germany), identification of K. pneumoniae isolates was performed 19. Verified isolates of K. pneumoniae were stored at -20ºC in TSB medium (Merck, Germany) with 15% glycerol for next steps. This study was approved by the ethical committee of Yasuj University of Medical Sciences.
Antibiotic susceptibility testing (AST): Susceptibility of K. pneumoniae isolates was determined by the disk diffusion method according to the Clinical and Laboratory Standards Institute (CLSI) guidelines 20. Twelve antibiotic disks (MAST, UK) including amoxicillin/clavulanic acid (AMC; 30 µg), cephalothin (CEP; 30 µg), ceftazidime (CAZ; 30 µg), imipenem (IMP; 10 µg), chloramphenicol (CLR; 30 µg), nitrofurantoin (NI; 300 µg), tetracycline (TET; 30 µg), gentamycin (GEN; 10 µg), amikacin (AMI; 30 µg), ciprofloxacin (CIPR; 5 µg), nalidixic acid (NAL; 30 µg), and trimethoprim-sulfamethoxazole (SXT; 25 mg) were used. K. pneumoniae ATCC 700603 was used as the control for antibiotic resistance. MDR was outlined as acquired non-susceptibility to a minimum of one agent in 3 or more antimicrobial classes 21.
DNA extraction: The boiling method with some modification was used to extract the genome of bacteria 22. In short, 0.5 McFarland bacterial suspension was prepared first, and then 300 μl of it was transferred to the 1.5 ml microtube containing sterile distilled water. The suspension within the microtube was homogenized with the vortex and then boiled for 10 min at 100°C in a water bath. In the next step, microtube was centrifuged for 10 min at 14000 g and the supernatant containing genome was transferred to the 0.5 ml microtube and kept at 18°C until the next step.
PCR for detection of integron genes: Polymerase Chain Reaction (PCR) was carried out for detection of integron genes (intI1, intI2, and intI3) using the primers described in table 1 23,24. Each reaction mixture in a final volume of 25 µl contained 12.5 µl of 2X Master mix (Amplicon, Denmark), 0.3 μl of each primer (20 pM/μl), 2.5 µl of DNA template and 9.4 μl of distilled water.
The planned conditions in the thermocycler (BioRad, T100, USA) for amplification of each of the genes examined were as follows; initial denaturation at 94°C for 4 min; 35 thermal cycles of denaturation at 94°C for 40 s, annealing at 58°C (For intI1 genes) or 51°C (For intI2 and intI3 genes) for 30 s, and extension at 72°C for 40 s; and a post-PCR final incubation at 72°C for five minutes. The amplified PCR products were electrophoresed on 1.5% agarose gel containing 0.5 μg/ml ethidium bromide and visualized under a UV transilluminator (Major science, Taiwan).
Characterization of gene cassettes inserted in the variable regions of class 1 integrons: PCR was carried out for amplification of variable regions of class 1 integron similar to the planned conditions for the detection of intI1 gene, except that the annealing temperature was 60°C using primers described in table 1 25. For each integron-positive isolate, some of the PCR product was electrophoresed and, after ensuring that the expected fragment was present, the remaining PCR product was sent to Macrogen company (Seoul, South Korea) for sequencing.
Obtained sequences were submitted in the NCBI database and blasted by online BLAST search (http://www.ncbi.nlm.nih.gov/BLAST/).
Statistical analysis: Data were analyzed with SPSS software (Version 15, Chicago, IL, USA). The chi-square and Fisher’s exact tests were used for determining the association between presence of intI genes and antibiotic resistance status. A p<0.05 was regarded statistically significant.
Results :
Antibiotic susceptibility test results: Among 196 studied isolates of K. pneumoniae, 137 isolates (69.9%) were collected from females and the remaining 59 isolates (30.1%) were obtained from male patients. The results of the AST showed that resistance to amoxicillin is highest (38.8%). Resistance rate to other antibiotics was as follows; they were cephalothin (32.1%), nitrofurantoin (22.4%), ceftazidime (12/8%) trimethoprim-sulfamethoxazole (9.7%), tetracycline (8.7%), gentamycin (5.1%), nalidixic acid (3.6%), chloramphenicol (3.6%), ciprofloxacin (2.6%), and amikacin (1%). There was not any imipenem-resistant isolate. Twenty-eight (14.3%) isolates were MDR and 39 different antibiotic resistance profiles were observed (Table 2).
Detection of integrons and characterization of gene cassettes: Among the 196 K. pneumoniae isolates, class 1 integron (intI1) was identified in 24 (12.2%) isolates while intI2 and intI3 genes were not found in any of the studied K. pneumoniae isolates. In table 2, the frequency of intI1 gene-positive isolates in each of the resistance profiles is shown.
Significant differences were observed between susceptibility patterns of trimethoprim-sulfamethoxazole, tetracycline, amoxicillin/clavulanic acid, ceftazidime, ciprofloxacin, chloramphenicol, nalidixic acid, cephalothin, gentamycin and presence of class1 integron in K. pneumonia isolates (Table 3). While 17 out of 28 (60.7%) MDR K. pneumoniae isolates harbored the intI1 gene, only 4.2% of non-MDR isolates carried intI1 gene (p<0.001).
Eighteen out of 24 (75%) intI1 positive K. pneumoniae isolates were found to have 7 different gene cassette arrays including the dfrA5, dfrA25, dfrA7, aadA1, dfrA17-aadA5, dfrA1-orfC and aadB-cat-blaOXA10-aadA1 (Table 4, Figure 1). In the remaining 6 intI1 positive K. pneumoniae isolates, variable region was not detected.
Among seven different cassette arrays found in K. pneumoniae, aadA1 was the most frequent gene cassette detected in 9 (37.5%) out of 24 intI1 positive isolates. dfrA5, dfrA25, dfrA7, dfrA1-orfC and aadB-cat-blaOXA10-aadA1 gene cassette arrays were found in one isolate. Four isolates were found to be positive for dfrA17-aadA5 gene cassette array.
Discussion :
Similar to previous reports from Iran and other countries, all of the studied K. pneumoniae isolates were found to be susceptible to imipenem 26-30. Also, in our previous study, only 1% of urinary E. coli isolates were imipenem resistant 22. The low and prudent administration of imipenem by physicians in Iran and consequently reducing selection pressure of this antibiotic on bacteria has made it the most reliable and most effective antibiotic against bacteria such as E. coli and K. pneumoniae. In the present study, the rate of resistance to trimethoprim-sulfamethoxazole (9.7%), tetracycline (8.7%), gentamycin (5.1%), nalidixic acid (3.6%), chloramphenicol (3.6%), ciprofloxacin (2.6%) and amikacin (1%) all were less than 10%. These relatively low rates were not observed in other studies from Iran and other countries in recent years. The resistance rate of urinary K. pneumoniae isolates in a study by Akram et al in India was reported as co-trimoxazole 53%, tetracycline 53%, gentamycin 53%, ciprofloxacin 47%, and amikacin 35% 31. Also, in another study in India by Mariya and Hatkar, the rate of resistance to co-trimoxazole and amikacin was 59.73% and 31.95%, respectively 29. In another study in Iran, the resistance rate of K. pneumoniae isolates was reported as co-trimoxazole 95.3%, tetracycline 64.7%, gentamicin 76%, chloramphenicol 57.3%, ciprofloxacin 43.3%, and amikacin 50.7% 32. Differences in the pattern of antibiotic resistance in different countries and even in different regions of a country may depend on different factors such as rate of access and use of different antibiotics, population, climatic conditions and public health levels 33.
In the present study, the highest rate of antibiotic resistance was observed for amoxicillin/clavulanic acid in 38.8% of K. pneumoniae isolates. Unfortunately, in Iran, the arbitrary use of this antibiotic among people has a very high prevalence that increases the selective pressure of the antibiotic on bacteria leading to resistance mechanisms.
In addition to amoxicillin/clavulanic acid, the resistance rate of our isolates was found to be higher than 10% for only three other antibiotics including cephalothin (32.1%), nitrofurantoin (22.4%) and ceftazidime (12/8%).
In the present study, class1 integron was detected in 12.2% of K. pneumoniae. This frequency is much lower than that reported by Ahangarzadeh Rezaee et al in northwest Iran (78%) 32. Considering that in the study of Ahangarzadeh Rezaee et al, 99.3% of isolates were MDR and in our study only 14.3% of isolates were MDR, this high difference between the presence of the integron in these two studies was not surprising. The above comparison may illustrate well the association between the emergence of MDR bacteria and the presence of integron. Also, Li et al and Lina et al reported a higher prevalence of class I integron compared to the current study 4,26. The prevalence of class I integron in their studies was 51.1% and 54%, respectively. On the other hand, in another study in Iran by Seyed Javadi et al, the prevalence of class I integrons among K. pneumoniae isolates (13.3%) was very close to our study 34.
Similar to other studies class 2 and class 3 integrons were not found in any of our K. pnemoniae isolates, while Ahangarzadeh Rezaee et al reported class 2 integrons in 13.4 % of their isolates 25,32,35,36.
Significant differences were observed between the presence of the intI1 gene and susceptibility patterns of all antibiotics except amikacin and nitrofurantoin. Similar to our study, Li et al did not find a significant association between resistance to nitrofurantoin and the presence of class1 integron 4.
In the present study, 7 different cassette arrays were found in 75% of intI1 positive K. pneumoniae isolates. Among them, aadA1 was the most frequent gene cassette detected in 37.5% of 24 intI1 positive isolates. The aadA type gene cassettes confer resistance to aminoglycosides such as streptomycin that was widely used for treating UTIs in the early years 37. Since streptomycin usage has been limited to some specific human diseases, its frequent application in agriculture and food animals, especially in livestock production, may bring development of resistance via acquiring resistance gene cassettes 37. Hence, this gene cassette can remain in animal pathogenic strains of E. coli and be disseminated to other strains by horizontal gene transfer mechanisms.
The high prevalence rate of gene cassette arrays with dfr genes in the current study, which codes for Dihydrofolate Reductase (DHFR) conferring resistance to trimethoprim, can reflect the wide use of trimethoprim in the treatment of urinary tract infections in recent years.
Similar to findings of previous studies 4,19, in the present work, 25% of K. pneumoniae isolates were found to carry class1 integron without gene cassettes. In a study from China performed on K. pneumoniae isolates, dfrA1-orfC was the most predominant cassette array among 10 different identified cassette arrays among which dfrA5, aadA1 and dfrA1-orfC gene cassettes were also detected in our study 4. Also, Salimizand et al identified only 3 gene cassettes or cassette arrays in K. pneumoniae isolates including arr-5, aacA4-orfD, and dfrA17-aadA5, among which the latter cassette was found in our study 27.
In the present study, all isolates with the dfrA7, dfrA17-aadA5, and dfrA1-orfC gene cassette arrays exhibited a resistance phenotype to trimethoprim sulfamethoxazole. Two isolates with dfrA25 and dfrA5 cassettes, contrary to our expectation, have not shown resistance to trimethoprim sulfamethoxazole. The reason for this can be found in the presence of weak promoters or the presence of mutations in the region between the two specific sequences of promoters 38.
The cat gene that codes for Chloramphenicol Acetyl Transferase (CAT) conferring resistance to chloramphenicol was found in aadB-cat-blaOXA10-aadA1 cassette array. Although this gene cassette array has already been reported from China 39, identification of this gene cassette array in K. pnumoniae isolates was reported for the first time in Iran. Surprisingly, in the current study, this cassette array was found in one chloramphenicol sensitive isolate and despite the presence of cat cassette, a large distance between the promoter and the corresponding gene cassette may affect the transcription of cat gene in this isolate 38. blaOXA10 gene found in aadB-cat-blaOXA10-aadA1 cassette array encodes for β-lactamase enzymes conferring resistance to beta-lactam antibiotics such as amoxicillin and ampicillin 40.
Conclusion :
In the present study, the proportional prevalence of integrons and the antibiotic resistance among K. pneumoniae isolates clearly shows the importance of integrons in dissemination of antibiotic resistance. aadA1 was found to be the most prominent gene cassette among 7 different cassette arrays identified in K. pneumoniae isolates. Continuous monitoring and characterization of integrons and their associated gene cassettes could be helpful in controlling the rate of antibiotic resistance by planning to take preventive measures to hinder the spread of resistant strains.
Acknowledgement :
This study has been supported by Deputy of Research and Technology, Yasuj University of Medical Sciences.
Conflict of Interest :
The authors declare no conflicts of interest.
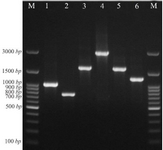
Figure 1. PCR assay for identification of some gene cassette arrays among K. pneumoniae isolates. Lane M: DNA marker, lane 1: aadA1 (1000 bp), lane 2: dfrA7 (750 bp), lane 3 and 5: dfrA17-aadA5 (1650 bp), lane 4: aadB-cat-blaOXA10-aadA1 (3000 bp), lane 6: dfrA1-orfC (1300 bp).
|

Table 1. Primers used for the detection of class 1-3 integrons and variable region of class 1 integrons
|
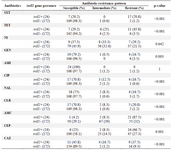
Table 2. intI1 gene presence status in K. pneumoniae isolates according to antibiotic susceptibility pattern
SXT=Trimethoprim-Sulfamethoxazole, TET=Tetracycline, NI=Nitrofurantoin, GEN=Gentamycin, AMI=Amikacin, CIP=Ciprof-loxacin, NAL=Nalidixic acid, CLR=Chloramphenicol, AMC=Amoxicillin/Clavulanic acid, CEP=Cephalothin, CAZ=Ceftazidime.
|
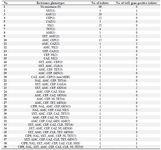
Table 3. Frequency of intI1 gene-positive isolates in each of the resistance profiles
|
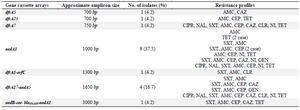
Table 4. Gene cassette arrays found in class 1 integrons and related resistance profiles
|
|