Glycation Inhibition of Bovine Serum Albumin by Extracts of Momordica charantia L. using Spectroscopic and Computational Methods
-
Oso, Babatunde
-
Department of Biochemistry, McPherson University, Seriki Sotayo, Ogun State, Nigeria
-
Agboola , Olubukola
-
Department of Biochemistry, McPherson University, Seriki Sotayo, Ogun State, Nigeria
-
 Olaoye , Ige
Department of Biochemistry, McPherson University, Seriki Sotayo, Ogun State, Nigeria, Tel: +234 08065056590; E-mail: igelaoye@gmail.com
Olaoye , Ige
Department of Biochemistry, McPherson University, Seriki Sotayo, Ogun State, Nigeria, Tel: +234 08065056590; E-mail: igelaoye@gmail.com
Abstract: Background: Momordica charantia (M. charantia) has been used in traditional medicine for the management of complications associated with diabetes mellitus. Several phytochemicals with different pharmacological properties have been previously identified from the botanical; however, the mechanisms of actions of this plant vis-à-vis inhibition of non-enzymatic protein glycation are not known. This study aimed at understanding the putative mechanisms underlying the antiglycation properties of M. charantia extracts experimental and theoretical approaches.
Methods: The antiglycation properties of the plant were evaluated by studying the inhibitory actions of methanol and aqueous extracts on glucose-induced glycation of Bovine Serum Albumin (BSA) and protein aggregation. The mode of binding of identified phenolics of the botanical with BSA, amyloid beta-peptide (1-42) and 3D amyloid beta (1-42) fibrils were also investigated.
Results: The in vitro experimental properties of the extracts showed that the extracts could prevent inductions of protein glycation and protein folding. The molecular docking analyses revealed that phenolics had better binding affinities with chlorogenic acid showing the highest binding score (-7.13±0.04 kcal/mol) towards BSA than glucose and their respective interactions with BSA could prevent glucose-induced protein aggregation.
Conclusion: Consequently, the results of this study provide insight into the probable mechanisms of actions of the extracts of M. charantia against the inhibition of advanced glycation end products formation.
Introduction :
Glycation is a non-enzymatic, spontaneous reaction between biomolecules and sugars (glucose, fructose and ribose) leading to the formation of Advanced Glycation End products (AGEs) 1-3. Over the past three decades, several research work has been carried out on glycation and anti-glycation processes in diabetes, due to the fact that excess glucose in the blood can cause the free sugars to bind with biomolecules 4,5. Therefore, agents with the ability to inhibit oxidative stress and glycation will be of great significance in the management of diabetes. Traditionally, plant extracts play vital roles in the management of diabetes and have been gaining recognition 6. Several studies have revealed the hypoglycemic efficiency of crude extracts from plants and their bioactive compounds 7. The ability of such plants to inhibit Advanced Gycation End products (AGEs) is in fact gaining popularity in the treatment of diabetes without the control of the level of blood glucose 8,9.
Momordica charantia L. (M. charantia), commonly called bitter gourd or bitter lemon and belonging to a family of Cucurbitaceae, has been reported to have significant pharmacological effects especially the leaves 10, fruits and seeds 11. Also, it has been used for the management and treatment of various ailments such as bacterial and viral infections, obesity, and cancer 12. Other pharmacological significances of the plant are antimalarial, antihelmintic, contraceptive, laxative and abortifacient. These pharmacological potentials of the botanical have been attributed to the high contents of phenolic acids, flavonoids, isoflavones etc. 13. Also, the work of Piwowar et al 14 identified phenolic acids such as chlorogenic acid, ferulic acid as possible antiglycoxidative agents. The interaction of these phytochemicals with suitable drug targets can be explained using different predictive studies. This can be achieved by using various computational models which have been helpful in the discovery of drug-like compounds and validation of drug targets 15. Therefore, this study assessed the in vitro inhibitory effects of methanol and aqueous extracts of dried leaf of M. charantia L. on albumin glycation properties. The study also analysed the binding behaviour of the bioactive components of the leaf with Bovine Serum Albumin (BSA) and amyloid beta-peptide and amyloid beta fibrils.
Materials and Methods :
Chemicals: All chemicals used were of analytical grade. BSA and Congo red are products of Kem Light, India, Methanol, sodium dihydrogen phosphate, and disodium hydrogen phosphate were products of Guangzhou JHD Chemical Reagent Co., Ltd. Guangzhou, China.
Sample preparation: The plant extract sample was prepared following the method described by Ofuegbe et al 16 with slight modification. Fresh leaves of M. charantia were air-dried at room temperature of 29±1°C. The sample was authenticated at the Laboratory of Biological Science, McPherson University, Nigeria. The dried leaves were pulverized and divided into two. Exactly 500 g of pulverized sample from one division was extracted with 1 L of methanol at room temperature of 29±1°C for 72 hr and later filtered. In a separate beaker, 500 g of pulverized sample from the second division was extracted with 1 L of distilled water at room temperature of 29±1°C for 72 hr and later filtered. The crude extracts were used as the corresponding extracts for the subsequent analyses.
In vitro glycation of BSA: The procedure for the preparation of glycated albumin was according to the method of Safari, Sheikh, and Kashani 17 with minor modifications. Briefly, BSA (0.15 g/ml) was incubated in phosphate buffer (0.1 M, pH=7.4, containing 0.01% sodium azide) containing D-glucose (18 mg/ml) as glycated group. The control group had the same concentration of BSA prepared in phosphate buffer (0.15 g/ml) without D-glucose. To study the influence of the extracts on BSA glycation, different concentrations of the extracts (50, 100, 150, and 200 µg/ml) were added to the incubation medium containing BSA (0.15 g/ml) and D-glucose (18 mg/ml) in phosphate buffer (0.1 M, pH=7.4, containing 0.01% sodium azide). The solutions were incubated at 37°C for 72 hr. The reaction mixtures were then dialyzed against phosphate buffer (0.01 M, pH=7.4) for 24 hr to remove unbind glucose from the samples.
Measurement of glycated BSA: The degree of BSA glycation was estimated through colourimetric method using Thiobarbituric Acid (TBA) 18,19. In principle, glycated BSA is hydrolyzed by oxalic acid to produce 5-hydroxymethyl furfural (5-HMF) which reacts with TBA to form a chromophore with a maximum absorbance at 443 nm. Briefly, 2 ml of 20% Trichloroacetic Acid (TCA) was added to 4 ml of glycated sample and centrifuged for 10 min at 3000 rpm using 800D Centrifuge Machine. Exactly, 1 ml phosphate buffer and 0.5 ml oxalic acid (0.3 N) were added to the sediment and boiled in the water bath for 1 hr. After cooling, 0.5 ml of 40% TCA was added to each sample and centrifuged (10 min, 3000 rpm) using 800 D Centrifuge Machine. 0.5 ml 0.05 M TBA was then added to 1 ml of supernatant and heated in 40°C for 30 min. The absorbance of each sample was taken at 443 nm using GS UV11 Spectrophotometer. The percentage inhibition of glycated BSA was calculated relative to the control according to the equation:
Percentage inhibition=A0-A1A0×100
A0 = absorbance of untreated BSA + glucose; A1 = Absorbance of sample (BSA + glucose with extract)
Determination of protein aggregation: Congo red was used to assess the effect of plant extract on glycation-induced BSA aggregation 20. Congo red binds to amyloid beta and forms a complex with maximum absorbance at 530 nm. Briefly, 0.5 ml of glycated BSA was incubated with 0.5 ml of Congo red (100 μM in phosphate buffer with 10% ethanol) at 25°C for 20 min. The absorbance of the sample was then recorded at 530 nm using GS UV11 Spectrophotometer. The percentage inhibition of protein aggregation was calculated relative to the control according to the equation:
Percentage inhibition=A0-A1A0×100
A0 = absorbance of untreated BSA + Glucose; A1 = Absorbance of sample (BSA + glucose with extract)
Molecular docking: The compounds selected for macromolecule-phytochemical interactions are phenolic acids previously reported by Lee et al 21. The phenolics are chlorogenic acid (PubChem CID: 1794427), gallic acid (PubChem CID: 370), ferulic acid (PubChem CID: 445858), p–coumaric acid (PubChem CID: 637542), and caffeic acid (PubChem CID: 689043). The 3D SDF structures of the selected phenolics and glucose (PubChem CID: 5793) were obtained from the PubChem database (www.pubchem.ncbi.nlm.nih.gov). The structures of BSA, amyloid beta-peptide (1-42) and3D amyloid beta (1-42) fibrils with PDB IDs 4OR0, 1IYT and 2BEG were retrieved from the protein databank (www.rcsb.org). The molecules were subsequently converted to the pdbqt format using Autodock tools. Blind docking of the ligands to various protein targets and determination of binding affinities was carried out using PyRx-Python Prescription 0.8 (The Scripps Research Institute) 22 with the grid set at (Centre: X: 35.1650; Y: 23.9873; z: 98.0448), (X: -0.6159; Y: -0.0652; Z: 11.2694), and (X: 0.4623; Y: 0.2758; Z: -8.9351), respectively with Dimension (Armstrong): (X: 144.9461; Y: 64.2626; Z: 88.7264), (X: 33.6613; Y: 22.3151; Z: 49.5333), and (X: 45.8883; Y: 22.9133; Z: 27.2084). The interactions were visualized through the Protein-Ligand Interaction Profiler server (https://projects.biotec.tu-dresden.de/ plip-web/plip). The binding affinities of compounds for the three protein targets were recorded.
Statistical analysis: The findings were evaluated using a one-way variance analysis (ANOVA) for mean differences between the different extracts followed by multiple comparison tests by Duncan for post-hoc correlations at p<0.05 and reported as a means±standard deviation of three determinations.
Results :
In vitro glycation of bovine serum albumin and determination of protein aggregation: The effects of aqueous and methanol extracts of M. charantia on inhibition of glucose-induced BSA-glycation is shown in figure 1. The results showed a significant increase in percentage inhibition of AGEs formation in the methanol extract at 50 μg/ml compared to the observable increase in the corresponding aqueous extract. The percentage inhibition of the formation of AGEs increased in a concentration-dependent manner. However, a significant increase in the percentage inhibition of the formation of AGEs by the aqueous extract was observed at a concentration of 200 μg/ml. In the determination of protein aggregation, a higher fold of inhibition of aggregation of albumin was observed in the methanol extract compared with the aqueous extract. Significant inhibitions of protein aggregation were observed at 150 and 200 μg/ml with p<0.05 (Figure 2).
Molecular docking: Although, while the ability of a protein to fold into well-defined tertiary structures and form amyloid fibrils could be a general property of protein, the propensity of any protein to form amyloid fibrils depends on its amino acid sequence and the protein concentrations 23,24. Thus, the probable binding mechanisms of the identified compounds were further assessed through molecular docking to investigate the binding affinities of the identified compounds with amino acid residues of bovine serum albumin and amyloid beta fragments. Chlorogenic acid showed the best binding score of -7.13±0.04 kcal/mol through its interaction with BSA using molecular docking analysis followed by ferulic acid and caffeic acid. Glucose had the least binding affinity with serum bovine albumin with the binding score of -5.55±0.15 kcal/mol. The chlorogenic acid and BSA complex formed one hydrophobic bond at ALA-193, five conventional hydrogen bonds at HIS-145, ARG-185, GLU-186, GLU-424, and SER-428, and one carbon–hydrogen bond at Gln189, and three salt bridges at ARG-427, LYS-431, and ARG-435. In contrast, the complex between BSA and Gallic acid was stabilized by one hydrophobic bond at ALA-290, five conventional hydrogen bonds at GLU-152, TYR-156, SER-191, ARG-198, ALA-290, and one salt bridge at ARG-198 position. Ferulic acid, p–Coumaric acid, and Caffeic acid interacted with BSA through hydrophobic bonds at positions LEU-189, and ALA-193, ALA-193, ILE-455, and ARG-458, and TYR-149, LEU-237, and ALA-290, respectively while their respective interactions through the conventional hydrogen bonds occurred at positions LEU-189, LYS-431, and ARG-435, SER-428 and ARG-435, and TYR-149 and ARG-256. Glucose interacted with bovine serum albumin at GLU-152, TYR-156, SER-191, ARG-198, ALA-290 (Table 1, Figure 3).
The results of the binding energies of the interactions of the selected compounds with amyloid beta-peptide (1-42) and amyloid beta (1-42) fibrils and their respective ligand-receptor binding pose are presented in tables 2 and 3. All the selected compounds were found to have good binding affinity against amyloid beta-peptide (1-42) and amyloid beta (1-42) fibrils. Chlorogenic acid had the most negative binding energies when interacted with the selected peptides followed by ferulic acid. There was no significant difference (p<0.05) between the interactions of ferulic acid, p–coumaric acid, and caffeic acid with amyloid beta-peptide (1-42). Gallic acid and ferulic acid interacted equally with amyloid beta (1-42) fibrils.
The 3D interactions between the selected compounds and the peptides are presented in figures 4 and 5. It was observed that chlorogenic acid directly interacted with residues of amyloid beta-peptide (1-42) through hydrophobic interaction at positions HIS-6 and TYR-10, stabilized by hydrogen bonding at GLU-3. VAL-12 was present in the interactions of amyloid beta-peptide (1-42) with gallic acid, ferulic acid, p–coumaric acid, and caffeic acid.
Gallic acid, ferulic acid, and caffeic acid formed a hydrogen bond with amyloid beta-peptide (1-42) at GLN-15 and LYS-16 residues. The bindings of ferulic acid and caffeic acid with amyloid beta-peptide (1-42) were further stabilized by salt bridges formed at LYS-16. Chlorogenic acid interacted with the amyloid beta (1-42) fibrils at residues ASN-27, LYS-28, and ALA-30 through conventional hydrogen bonds. LEU-17, PHE-19, and VAL-40 are predominant amino acid residues through which Gallic acid, ferulic acid, and caffeic acid established hydrophobic bonds with the amyloid beta (1-42) fibrils.
Discussion :
The investigated extracts of M. charantia were found to be active against the in vitro formation of protein glycation and induction of protein folding 25. The leaf of this plant has been reported to have diverse biological activities, such as antimicrobial, anti-inflammatory, and hepatoprotective properties 16. It is commonly used in folk medicine for the management of diabetes mellitus since it is alleged to possess hypoglycemic activity and prevent secondary complications associated with elevated serum glucose concentration 26. In this study water and methanol were selected as the extracting solvents because of differences in their polarities. Generally, methanol re sulted in higher biological activities relative to distilled water, although the aqueous extract resulted in higher inhibition of glucose-induced glycation of BSA at 200 μg/ml. This observation could be attributed to the transitional polarity of methanol which enhances the more effective partitioning of the polar and nonpolar phytochemicals trapped in the plant matrix. Thus, methanol could be suggested as being advantageous as an extraction solvent because of its extensive spectrum of polarity. The reported anti-AGEs activities vis-à-vis BSA glycation and protein aggregation could be responsible for the alleged premeditative effects on secondary complications of diabetes. Examples of major constituents of M. charantia include flavonoids and phenolics 27. These groups of phytochemicals have been employed as models for the production of different therapeutic agents suitable for the management of diabetes. Major phenolic acids are chlorogenic acid, ferulic acid, gallic acid, caffeic acid, and p-coumaric acid 21. Thus, the results obtained showed aqueous and methanol extracts of M. charantia could be used as a remedial agent to decrease AGEs formation in diabetes. To substantiate the revealed in vitro inhibitory potential of the aqueous and methanol extracts of M. charanta, the theoretical study was carried out and the result showed that all the investigated compounds had good interaction with higher binding scores based on their negative values towards BSA 28,29. Chlorogenic acid had the highest binding score (-7.13±0.04 Kcal/mol) towards BSA (Table 1). The high number of conventional hy
drogen bonds could be responsible for the highest binding score 30. This could imply that the pocket posed a tight interaction between chlorogenic acid with the protein molecule. However, the residues involved in the interactions depicted a different site other than the glucose binding site suggesting no competitive inhibition potential but a conformational change capability from chlorogenic acid towards BSA against glucose. Similarly, and in this order, the ferulic acid, caffeic acid and p-coumaric acid in almost similar binding sites but different from glucose binding site showed better binding scores (-6.15±0.15, -6.13±0.04 and -5.73±0.08 Kcal/mol, respectively) compared to glucose (-5.55±0.15 Kcal/mol). This trend of our molecular binding result is in agreement with the docking of caffeic acid and p-coumaric with BSA in Skrt et al, where caffeic acid exhibited a better binding score compared to p-coumaric. However, the lower binding score compared to chlorogenic acid could be due to lesser number of conventional hydrogen bond formation 30, while the number and type of hydrophobic interactions formed (Figure 3) by ferulic acid, p-coumaric acid and caffeic acid towards BSA could promote their higher binding score than glucose 15,31-33. Although all the investigated phenolic acids interacted with hydrophobic residues, none of them showed interaction with TRP-213 suggesting a possible different binding site when compared to protocatechuic aldehyde interaction as reported by Yanfang et al 34.
Interestingly, the gallic acid interacted with the BSA molecule with a better binding score at the glucose binding site. This higher binding score than glucose could be responsible for the better interaction exhibited by gallic acid. Also, the interaction of gallic acid towards BSA revealed a probable competitive inhibitory approach between gallic acid and glucose as seen in the number of conventional hydrogen linkages with similar residues (GLU-152, TYR-156, SER-191, ARG-198, ALA-290). In contrast, the better interaction of gallic could be due to the presence of hydrophobic interaction with ALA-290 and salt bridge formation with different residues (ARG-198) 15,31,32.
In addition, the formation of protein aggregation/fibrillation was examined using amyloid beta peptide (1-42) and amyloid (1-42) fibrils. The docking result showed that all the compounds interacted with the protein molecules. Chlorogenic acid had the best interaction with both molecules followed by caffeic acid while gallic acid showed the least interaction. This observation could be due to the highest binding score (-5.10±0.19 and -6.23±0.08 Kcal/mol) by chlorogenic acid for amyloid beta peptide (1-42) and amyloid (1-42) fibrils respectively (Table 2). The high binding affinity corresponds to the findings of Elokely and Doerksen 30 and Arthur and Uzairu 32 who reported the significance of conventional hydrogen bond and hydrophobic interaction, respectively. Surprisingly, all the compounds except chlorogenic acid interacted with VAL12, GLN15 and LYS16 via different bonds towards amyloid beta peptide (1-42) and LEU17 and VAL40 towards amyloid beta (1-42) fibrils (Figure 4).
Conclusion :
The crude aqueous and methanol extracts of M. charantia demonstrated potent inhibition of glucose-induced BSA glycation activity in in vitro. The mechanistic insight into the probable mechanisms of the alleged antidiabetic activities of the leaf of M. charantia also revealed the molecular interactions of some phytochemicals present in the plant with BSA could decrease the interaction of glucose with BSA and thus promote the inhibition of glucose-induced glycation. In addition, the putative interactions of some of the selected phytochemicals with amyloid beta and amyloid beta fibril showed the selected phenolic compounds could further alleviate the post-glycation effects of erroneously formed AGEs. Additional investigations are suggested on the pharmacological importance of this plant in order to establish its therapeutic potential against glycation induced disorders.
Acknowledgement :
The support provided by the staff of Department Biological Sciences, College of Natural and Applied Sciences, McPherson University, Seriki Sotayo, Ogun State, Nigeria is well appreciated.
Conflict of Interest :
The authors declare that they have no conflict of interest.
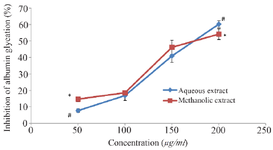
Figure 1. Anti-glycation properties of the extracts of M. charantiashowing the inhibition of albumin glycation. Each value stands for the mean±standard deviation of three determinations. Different symbols represent statistical difference at p<0.05 for each concentration.
|
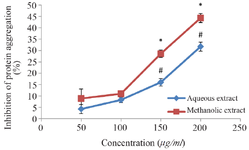
Figure 2. Inhibition of protein aggregation by extracts of M. charantia. Each value stands for the mean±standard deviation of three determinations. Different symbols represent statistical difference at p<0.05 for each concentration.
|
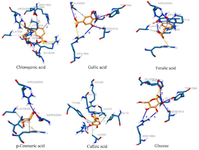
Figure 3. 3D interactions of the selected phenolics with bovine serum albumin.
|
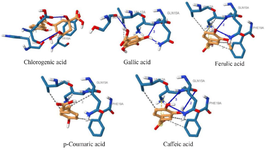
Figure 4. 3D interactions of the selected phenolics with amyloid beta-peptide (1-42).
|
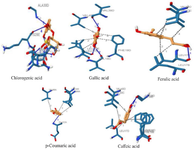
Figure 5. 3D interactions of the selected phenolics with amyloid beta (1-42) fibrils.
|
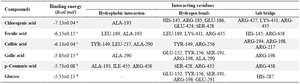
Table 1. Computed binding scores of the selected phenolics with bovine serum albumin
Different superscripts within a column represent statistical difference at p<0.05.
|
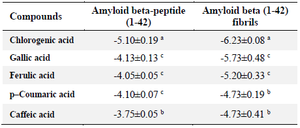
Table 2. Computed binding scores of the selected phenolics with amyloid beta-peptide (1-42) and amyloid beta (1-42) fibrils
Different superscripts within a column represent statistical difference at p<0.05.
|
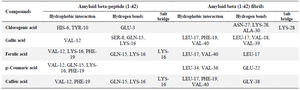
Table 3. Identified interaction sites of the selected phenolics with amyloid beta-peptide (1-42) and amyloid beta (1-42) fibrils
|
|